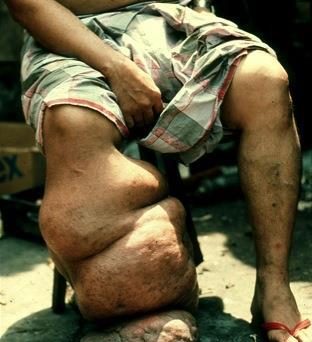
Onchocerciasis (river blindness) and lymphatic filariasis (elephantiasis) are neglected tropical diseases (NTDs) that cause severe disability and affect more than 106 million people, according to the latest reports from WHO.
These two NTDs are caused by parasitic roundworms, the filarial nematodes Onchocerca volvulus (onchocerciasis), Wuchereria bancrofti, Brugia malayi, and Brugia timori (lymphatic filariasis). These diseases have long-term chronic impacts on the health and wellbeing of endemic communities, including economic burden, disabilities, mental health issues and social stigmas.
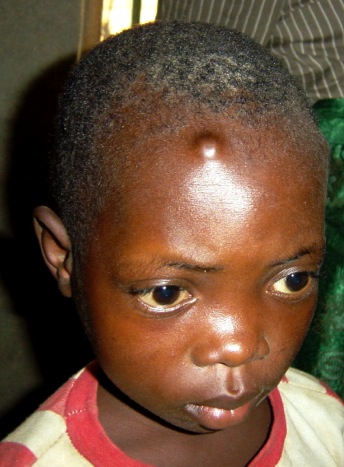
Global programmes for control and elimination, such as APOC and OEPA for onchocerciasis and GPELF for lymphatic filariasis have been developed and made significant progress, but existing approaches principally target only the early larval stage of filarial worms known as microfilaria. They require prolonged delivery with high treatment coverage to break the transmission cycle of the long-lived adult worm (6 – 8 years for LF and 10 – 12 years for onchocerciasis). The ‘holy grail’ for drug discovery and development in filariasis has been the search for a safe and effective drug that can target adult worms (macrofilaricide).
Our journey started over a decade ago after a breakthrough was made by Prof Mark Taylor and his co-workers that doxycycline could target the essential symbiont Wolbachia to deliver a safe macrofilaricidal cure clinically.
Targeting the symbiotic bacterium Wolbachia
Wolbachia are essential for multiple components of filarial nematode biology including larval growth and the production of offspring and, ultimately, survival of many filarial nematodes, including the causative parasites of onchocerciasis and lymphatic filariasis, making this symbiont a very attractive chemotherapeutic target for the treatment of both diseases.
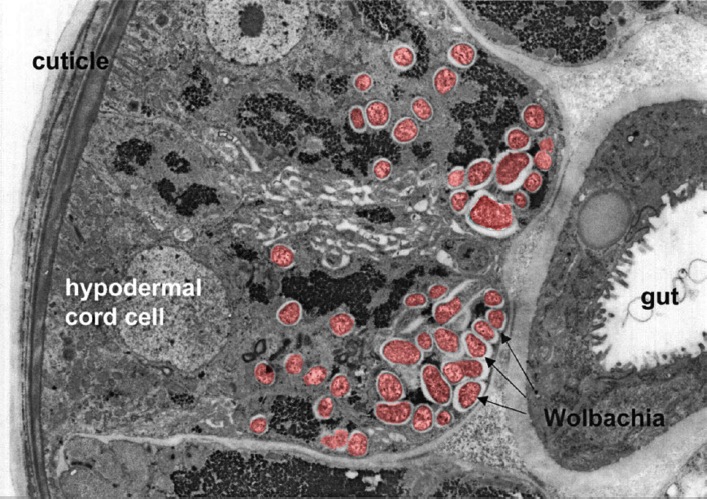
In addition, depleting Wolbachia from the parasitic worms leads to a slow innocuous death of the adult worms (over 12 – 20 months), which avoids the adverse effects of killing the adult worms too quickly.
Many of the current treatments that target microfilaria are not suitable for patients who co-infected with Loa loa (aka the ‘eye worm’) since killing the microfilaria of Loa loa may cause a severe adverse event. However, as Loa loa does not have Wolbachia within, macrofilaricides with specific anti-Wolbachia mode of action do not have such a drawback.
Although the macrofilaricidal activity of doxycycline through depletion of Wolbachia has been proven clinically in multiple trials, protracted treatment regimens (≥4-6 weeks) and contraindications (unsuitable for young children and woman of child bearing age) restrict its widespread implementation.
The A∙WOL discovery programme
Our anti-Wolbachia (A∙WOL) discovery and development programme was setup with a very clear aim: to find a new anti-Wolbachia treatment for onchocerciasis and lymphatic filariasis which would fulfil the following criteria:
1) An oral drug with short regimen (ideally ≤7 days).
2) Safe for children and women of child bearing age.
3) Affordable for the patients in the affected areas (mostly under-developed tropical countries).
To start with, we screened 10,000 chemical compounds selected from a commercial source (the BioFocus Soft-Focus library) using a phenotypic cell-based screen incorporating a Wolbachia-infected insect (Aedes albopictus) cell line [C6/36 (wAlbB)] to find new chemicals with anti-Wolbachia activity. We then selected one series of chemicals called thienopyrimidines as the starting point for a drug discovery and development project.
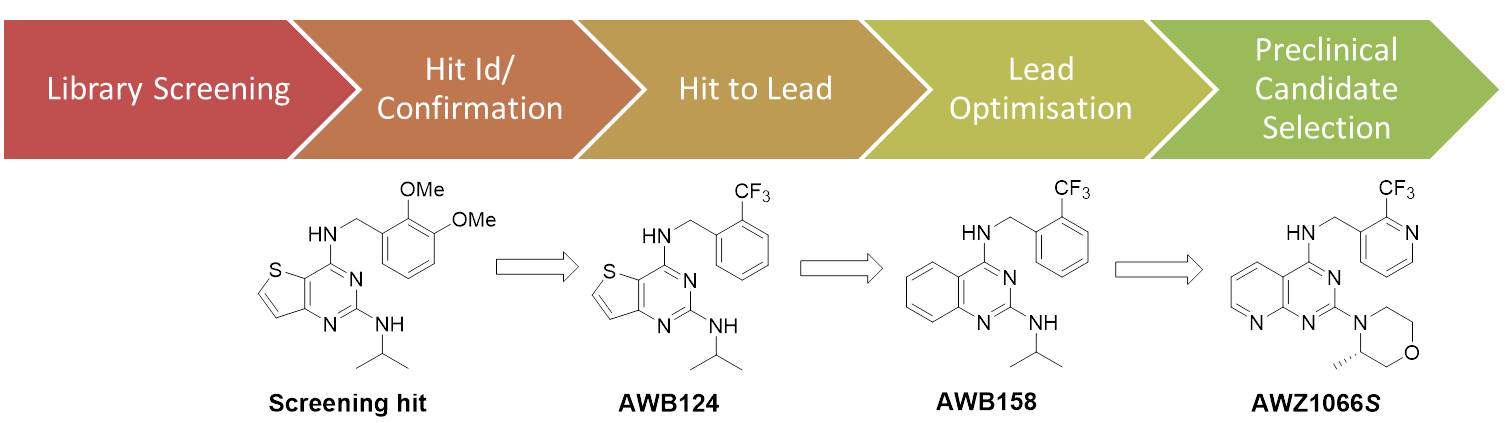
This project is a truly collaborative process relying heavily on toxicology/safety and drug metabolism/pharmacokinetic (DMPK) expertise from AstraZeneca and Eisai, medicinal chemistry expertise from University of Liverpool and Eisai and parasitology and pharmacology expertise from the Liverpool School of Tropical Medicine in order to deliver the shared goal of a high-quality drug candidate.
A candidate molecule discovered
After an extensive medicinal chemistry led, multi-parameter optimisation campaign, we have delivered the first novel synthetic candidate molecule coded AWZ1066S that can match all the required criteria set out in the programme and is undergoing preclinical development for the transition to human clinical trials.
AAWZ1066S showed far superior efficacy (depletion of Wolbachia and sterilisation of microfilaria production) than the ‘Gold-standard’ anti-Wolbachia drug, doxycycline, at 7-day treatment regimens in two independent filarial infection animal models. Importantly, AWZ1066S is highly specific against Wolbachia with little effect against a range of clinically relevant bacterial strains. This high specificity against Wolbachia minimises the impact on the gut microbiota, lowering the risk of dysbiosis (microbial imbalance) and the selection and emergence of resistance, in comparison to other anti-Wolbachia based broad spectrum antibiotic treatments.
Although the exact molecular target(s) of AWZ1066S is still unknown, we have obtained solid evidence to suggest that AWZ1066S has distinctive mechanism(s) that is different from many other known antibiotic anti-Wolbachia drugs that target either the protein or DNA synthesis pathways. In collaboration with Prof Ed Tate’s research group from Imperial College London, work has been initiated to uncover the mechanism of action of AWZ1066S. We anticipate that the target identification of AWZ1066S in Wolbachia will not only give further insight of how this molecule works, but also have potential to discover new biological pathway(s) in bacteria that have not been previously exploited.
Ultimately, we hope our research will make a significant impact on current global onchocerciasis and lymphatic filariasis elimination programmes that has the potential to dramatically reduce elimination time frames from decades to years.

Comments