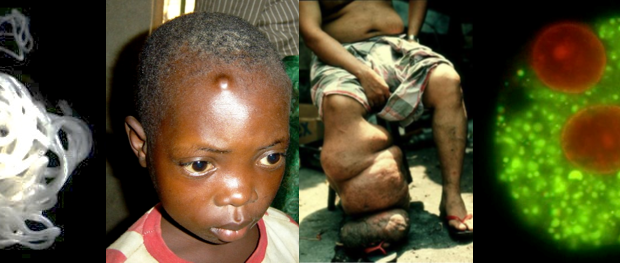
The filarial diseases, onchocerciasis (river blindness) and lymphatic filariasis (elephantiasis), disable over 106 million people throughout the tropics and as such, are a leading cause of global morbidity.
Current treatments for these diseases require long-term annual mass drug administration (MDA) to at-risk populations. Although there have been many success stories utilizing this approach, there are barriers to it delivering the elimination goals. These limitations include the risk of Serious Adverse Events (SAE) due to co-infection with the filarial worm Loa loa, potentially leading to encephalopathy, coma and death, the impact this fear has on adherence to treatment and achieving the necessary coverage, and the threat of drug resistance.
The primary aim of these MDA strategies is to interrupt transmission of the parasite by depleting the larval stages of the worms, the microfilariae (Mf), within humans.
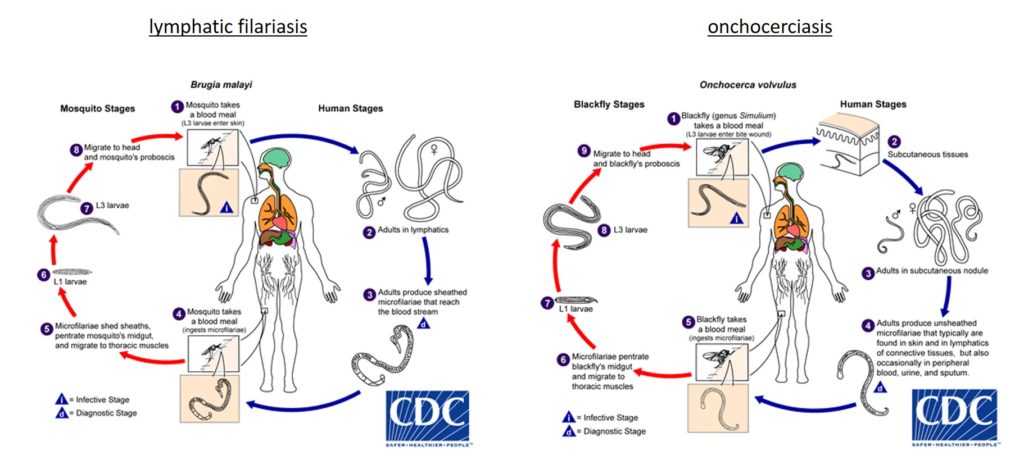
The Mf develop within biting insect hosts, which have ingested the Mf through a blood meal. Depleting these larval stages therefore limits the transfer of later stage larvae back into the human host where they develop to reproductive adults, thus completing the life cycle. What is lacking from this approach are drugs which can safely target the adult worms.
The anti-Wolbachia consortium
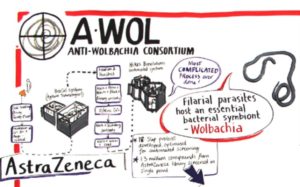
The anti-Wolbachia consortium (A∙WOL) has been focused on the discovery and development of curative treatments for these filarial diseases, through targeting the endosymbiotic bacteria Wolbachia within these parasitic worms. Clinical trials in humans have shown, killing the bacteria not only shortens the lifespan of these worms from decades to years, but also sterilize the worms. The latter is of great benefit as this permanently diminishes the larval pool of the life cycle, which is required for continued transmission. Death of the adults and halting the life cycle through sterilization leads to this curative treatment.
Although the consortium has proved the value of such treatment, current treatments are not suitable for MDA as it requires a 4 to 6 weeks course of treatment, which are contraindicated in children and pregnancy.
High-throughput screening
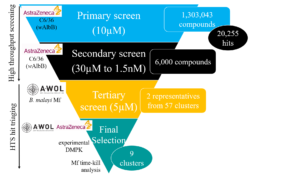
The partnership of A∙WOL with AstraZeneca enable the development of a truly industrial scale high throughput screen (HTS). This HTS was able to test 1.3 million compounds from AstraZeneca’s compound library, resulting in the identification of ~20,000 compounds with anti-Wolbachia activity.
This primary screen tested the compounds at a single dose. A secondary screen was carried out to test the hits in a dose response to quantify the potency of these drugs. Due to the vast number of hits identified, cheminformatics where used to focus on those with the best pharmacological properties and therefore likely to improve translation to drug candidates. This triaging included eliminating any compounds which had previously been tested by the A·WOL consortium, such as known antibacterials to enhance the chemical diversity of A∙WOL‘s portfolio.
Narrowing down the search
Following this secondary screen, bioinformatics was used again to cluster compounds into similar chemical structures. Samples from all of these clusters where taken forward into a tertiary screen, which measured the ability of these compounds to eliminate the symbiont from the worm. This screen identified 9 diverse clusters with potency against Wolbachia derived from Brugia malayi (nematode causing LF).
A step further was to test the speed at which the drugs eliminated the symbionts. Much to our surprise and delight 5 of these compounds showed the most rapid activity observed yet, eliminating the symbiont after only one day exposure. Only 1 other compound from the A·WOL drug portfolio has similar activity, AWZ1066S (as outlined in a recent BugBitten blog).
The excitement over this rapid killing action suggests a new mode-of-action, which is distinct from the generic protein synthesis inhibition of other A∙WOL drugs. Importantly, pre-clinical testing of these rapid killers, shows that this translates into shorter treatment periods of only a few days.
There is great anticipation as to how these 5 novel, fast acting compounds could act when carried forward into in vivo pre-clinical testing.

2 Comments