Chloroquine was first discovered in 1934 and for decades was widely used as a cheap and safe drug both to treat malaria, and as a prophylaxis. However, in the 1980s, widespread resistance developed in the parasite causing the most pathogenic form of malaria, Plasmodium falciparum. Understanding how resistance evolved took a long time and is initially based on knowledge of the mode of action of chloroquine.
Mechanism of chloroquine action
The red blood cell stage of the malaria parasite feeds on haemoglobin. Haemoglobin is transported to an organelle called the digestive vacuole where it is degraded, releasing amino acids and the toxic molecule, haem. This is biocrystallized to non-toxic haemozoin, which is insoluble and stays within the food vacuole.
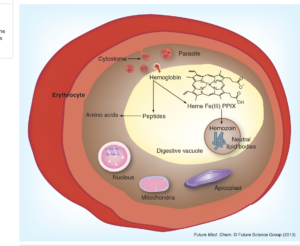
Disruption of this detoxification is the basis of the antimalarial action of chloroquine. Chloroquine diffuses into the digestive vacuole where the acidic conditions protonate the molecule, which inhibits its diffusion back out. As it accumulates, it binds to haem, blocking its biocrystallization, and this results in a build-up of toxic haem and results in the death of the parasite.
Piperaquine
Piperaquine, is another antimalaria drug that is structurally related to chloroquine and is now used in combination with dihydroartemisinin. Piperaquine also accumulates in the digestive vacuole and prevents the detoxification of haem to haemozoin. Unfortunately, resistance to piperaquine has been increasing in the last decade; spreading from Cambodia across Southeast Asia. Interestingly, piperaquine-resistant parasites have emerged that are more sensitive to chloroquine than piperaquine-sensitive ones, suggesting different resistance mechanisms are involved.
The P. falciparum chloroquine-resistance transporter (PfCRT)
In 2000 a report by David Fidock and colleagues associated chloroquine resistance with mutations to the gene for a digestive vacuole transmembrane protein, pfcrt. PfCRT is a member of the drug/metabolite transporter superfamily.
Mutant PfCRTs are thought to allow the efflux of chloroquine from the digestive vacuole, but whether this is the mechanism of piperaquine resistance was unclear.
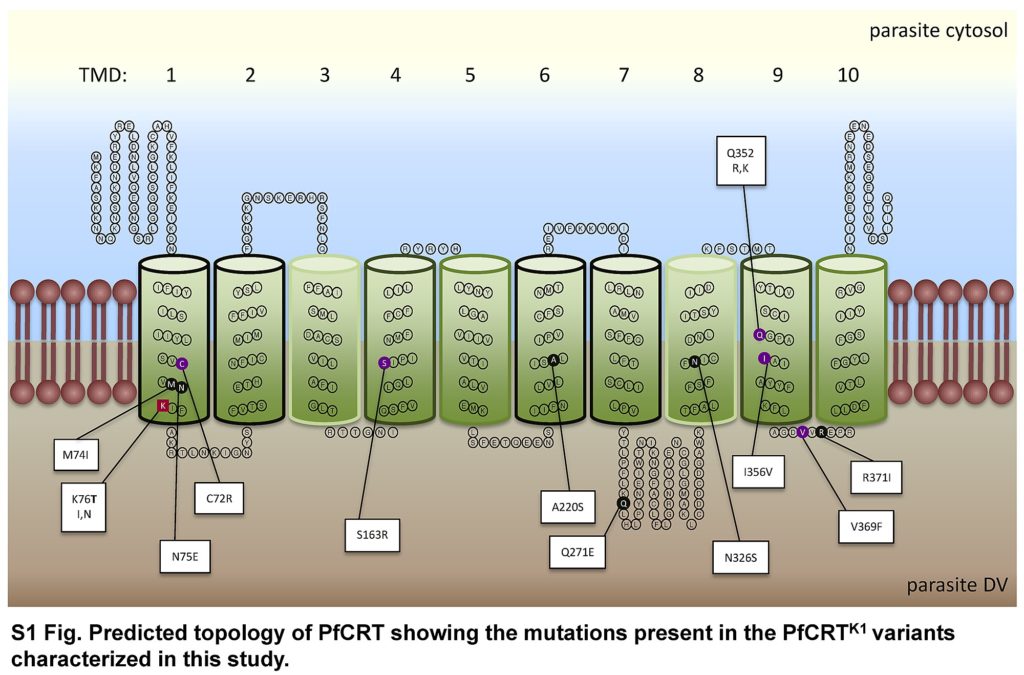
The structure of PfCRT
Until recently the detailed structure of PfCRT was unknown. However, with the aid of a new technique called cryo-electron microscopy, which can be used for membrane-embedded proteins, Jonathan King and colleagues have now resolved the structure of a PfCRT isoform of chloroquine-resistant, piperaquine-sensitive, South American 7G8 parasites. Because PfCRT is a very small molecule they first had to create an antibody fragment that would bind to it, increasing its size to enable resolution.
The team found that PfCRT consists of 10 helices that span the membrane of the digestive vacuole. They are arranged in 5 helical pairs with pairs 1-4 and 6-9 organised to form a central cavity that is wider on the digestive vacuole side. The central cavity has a net negative charge and has a maximum diameter capable of accommodating chloroquine or piperaquine. A positively charged lysine residue (K76), present in the PfCRT cavity of chloroquine-sensitive parasites, is thought to repel positively charged chloroquine and piperaquine, thus keeping these drugs within the digestive vacuole.
Resistant isoforms
Mutations resulting in amino acid substitutions associated with the chloroquine-resistant 7G8 isoform were mapped to residues that line the cavity of PfCRT. Mutations associated with piperaquine-resistant forms also line the cavity, although in different regions.
Chloroquine-resistant parasite all express one particular mutation, K76T. Here, the lysine residue is replaced by an uncharged threonine, which alters the distribution of the charge in the central cavity. In parasites with this mutation, chloroquine and piperaquine both bind to the altered cavity but only chloroquine can pass through and out of the digestive vacuole, reducing the inhibition of haemozoin formation.
The piperaquine resistance that emerged and spread from Cambodia may be the result of further mutations that gave rise to the Dd2 isoform.
PfCRT and resistance
To associate their structural studies with function, the group chose the F145I mutation found in the Dd2 isoform from Southeast Asia and the C350R mutation in the 7G8 isoform from South America; both conferred piperaquine resistance. Using expressed recombinant PfCRT variants of 7G8 containing these mutations they found a two-fold lower binding of piperaquine at a pH equivalent to the acidic digestive vacuole and increased transport rate, compared with the 7G8 isolates alone. In contrast, chloroquine transport was lower in these variants.
Biochemical and growth-inhibition comparisons of genetically modified 7G8 parasites expressing these variations showed that, when the Asian F145I and South American C350R mutations were introduced into the 7G8 isoform, piperaquine transport increased and chloroquine transport decreased thus piperaquine resistance increased and chloroquine resistance decreased.
Studies of electrostatic potential and molecular dynamics simulations showed that a reduction in the electronegativity of the cavity occurred in the 7G8-C350R variant as a result of an introduction of an arginine into the cavity which may reduce piperaquine binding, allowing it to be transported out. This was not the case for the 7G8-F1451 variant which, instead, may have caused a conformational change in the position of the helices of PfCRT, causing a change in cavity size and shape. It would thus appear that different mechanisms underlying piperaquine resistance and chloroquine re-sensitization have been selected in different geographical regions.
This comprehensive study has used a multitude of methods to shed light on the complex and varied mechanisms by which drug resistance has developed in P. falciparum. The authors rightly suggest that their research should be extended as a means of identifying combinations of drugs that act on this drug transporter in different ways.

Comments