
Bird eye worm
Oxyspirura petrowi is a heteroxenous nematode infecting birds in the United States (US). The eyeworm has been identified in numerous species in the orders Galliformes and Passeriformes, such as the lesser-prairie chicken and northern bobwhite quail (also known as bobwhite).
Oxyspirura petrowi is typically found on the eye surface under the nictitating membrane, as well as in the lacrimal ducts and other eye glands. Infections are associated with inflammation of the lacrimal ducts, keratitis and lesions on the Harderian gland.
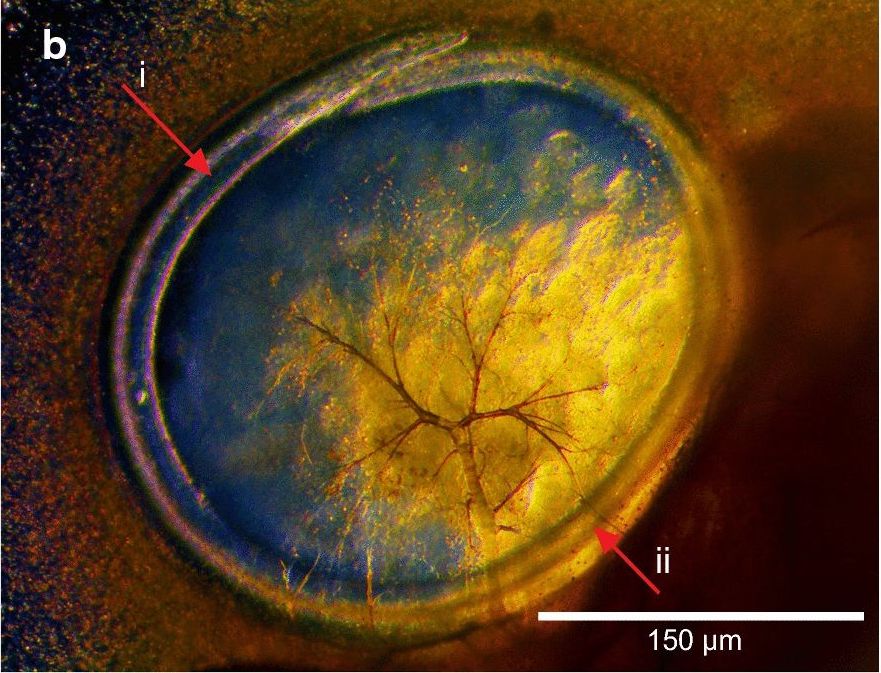
As the Harderian gland is associated with immune function, O. petrowi may negatively affect the host. Further, phylogenetic analyses have shown that O. petrowi is closely related to the eye worms Loa loa and Thelazia callipaeda, which can cause impaired vision and ulcerative keratitis.
Bobwhite and eye worm: a prevalent but elusive interaction?
The Rolling Plains Ecoregion of West Texas is reportedly the epicenter of O. petrowi infection in bobwhites. The parasite is highly prevalent here, with studies reporting prevalence’s of 58.7% to 100% in adult bobwhites.

Bobwhite populations have declined and there is concern that O. petrowi infection may be contributing to this. The pathological impact of infection alongside reports of bobwhite flying into stationary objects suggest impaired vision may occur in infected birds. However, little is known about the parasite life cycle and transmission dynamics.
Oxyspirura petrowi requires an intermediate host and molecular studies suggest grasshoppers, cockroaches and crickets may function, although a complete study of the parasite life cycle has not been conducted. Previous experiments achieved low (16.6%) infection rates in house crickets and complete development of O. petrowi was not observed.
A recent study by Kalyanasundaram and colleagues expanded upon previous research by providing a more effective means of infection and detailed examination of each stage of the O. petrowi life cycle in both crickets and bobwhite.
Establishing the parasite life cycle
Two aquariums were used to house crickets for experimental infection with O. petrowi (colony A) and an untreated control colony (colony B). Oxyspirura petrowi eggs were collected from gravid worms recovered from the eyes of wild bobwhite and fed to crickets. Crickets were dissected at specific intervals to monitor the development of O. petrowi.
Thirty adult pen-raised bobwhite were individually housed, with 25 assigned to the experimental group receiving crickets from colony A and 5 to a control group receiving crickets from colony B.
The maturation of O. petrowi in bobwhite was monitored through fecal floats (used to detect embryonated O. petrowi egg shedding, indicating life cycle completion) and bobwhite necropsies at specified intervals. PCR was used to analyse DNA from the larval stages of O. petrowi.

Bobwhite and crickets: a host system for an eye worm?
After 42 and 51 days of the study researchers successfully infected crickets (n= 45) and bobwhite (n= 25), respectively, with O. petrowi at a rate of 96%. All parasite life cycle stages were confirmed using morphological and molecular techniques.
A total of 113 O. petrowi larvae were identified in colony A crickets and infections ranged from 0–4 larvae per cricket with an average (± standard deviation [SD]) of 2.51 ± 2.4. All first-stage larvae were located in the cricket intestinal tracts, while the second-stage larvae were found fused to the cricket Malpighian tubules and intestines. Morphologically similar third-stage larvae were first recorded in the intestinal region. All stages of O. petrowi recovered from crickets were confirmed with PCR.
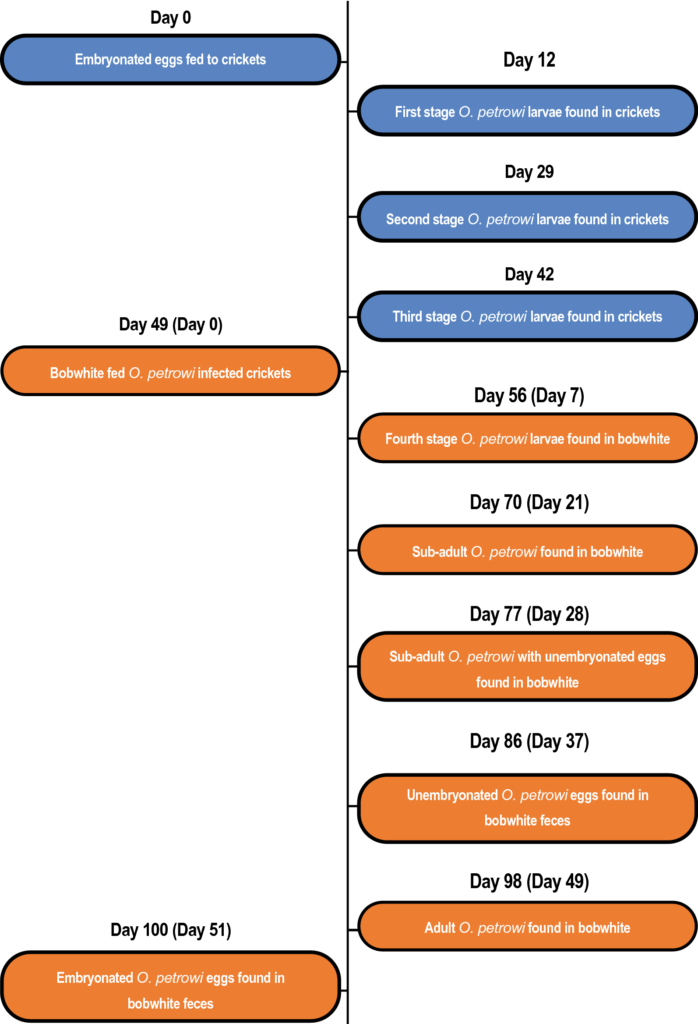
A total of 64 O. petrowi were recovered from the bird eyes and infection ranged from 0–6 worms per bird with an average (±SD) of 2.56 ± 1.5. Fourth-stage larvae were identified after 7- and 14-days post-exposure (DPE), but the reproductive organs were more developed by DPE 14. By DPE 21 worms were classified as sub-adults, and unembryonated eggs were found within the female oviducts by DPE 28.
The first adult O. petrowi were recovered from bobwhite on DPE 49. Finally, embryonated eggs were found within the feces of experimentally infected birds on DPE 51, indicating completion of the O. petrowi life cycle. All stages of O. petrowi recovered from bobwhite and bobwhite feces were confirmed with PCR.
Conclusions
By tracking the developmental progress within both the intermediate and definitive host, the experiments undertaken by Kalyanasundaram and colleagues provide a clearer understanding of the O. petrowi life cycle. This is thought to be the first study to replicate and monitor the complete O. petrowi life cycle in vivo.
In addition to providing an experimental model to examine the effects of infection on the host, a greater understanding of the parasite life cycle may enable better estimates into the potential for future epizootics of O. petrowi in bobwhite.

Comments