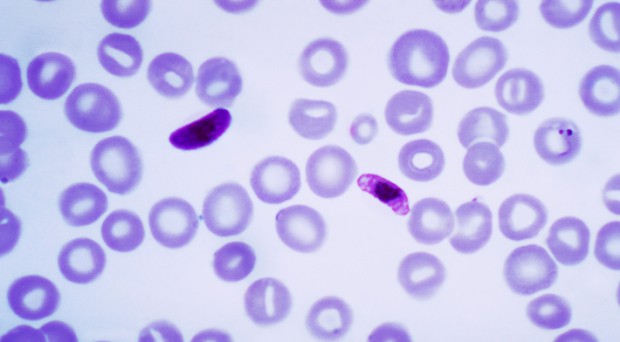
One of the most fascinating things about parasites is the complexity of their life histories. Some of the most complex of all are the malaria parasites. At different stages they can parasitize mosquitoes or vertebrates, live intra or extra cellularly and multiply asexually or sexually. Transition between these different stages requires switching on or off different sets of genes, a function performed by transcription factors (proteins that attach to a specific sequence of DNA next to the gene they control). Recent work has provided a greater understanding of one of these transcription factors thought to be the master switch involved in the formation of the sexual stages in the life cycle of a malaria parasite.
Malaria in the red blood cell
Once the malaria parasite invades a red blood cell (RBC) there are two possible ways in which it can develop: to divide asexually to produce schizonts, which will escape as merozoites and invade new RBCs, or to become either a female or several male sexual stages, known as gametocytes.
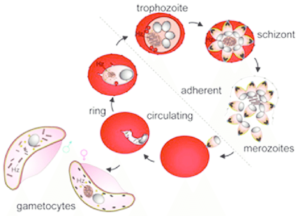
If schizonts and then merozoites are produced, the number of infected cells the host’s blood contains will increase. However, the malaria parasite will only be transmitted to the mosquito stage in its life cycle if gametocytes are produced. In the mosquito midgut, male gametes will fertilise females and the resulting zygote has the potential to traverse the midgut wall and develop into the next stage in the parasite’s life cycle.
Transcription factor AP2-G
Although the molecular mechanisms triggering one or other of these two developmental pathways are poorly understood, a transcription factor called AP2-G is known to be necessary to initiate the pathway that produces gametes in many species of malaria. This finding featured in a BugBitten blog in 2014 and is covered in detail in an excellent review.
A team of researchers based at the Universities of Glasgow, Edinburgh and Vermont and the Wellcome Trust Sanger Institute, Cambridge, has been using a rodent malaria, Plasmodium berghei, to shed more light on the role of this factor in the early stage of the development of malaria sexual stages.
In their study, recently reported in Nature Microbiology, they introduced two gene expression modules into a parasite line that already contained a third module that produced green fluorescence in male gametocytes and red fluorescence in female gametocytes. One of the modules was activated when rapamycin was added to the culture medium or given to mice for in vivo growth of the parasites (details are given in their paper). Their ingenious strategy produced two populations of parasites; one that over expressed ap2-g and produced gametocytes but not schizonts and one in which the ap2-g gene was effectively knocked out, giving a population that produced entirely schizonts.
Timing of commitment to sexual reproduction
The researchers were interested in assessing when this rodent malaria parasite became sensitive to programming for sexual development, because earlier work had shown that, in a human malaria parasite, Plasmodium falciparum, RBC stages become committed to becoming gametocytes when in the RBC cycle before the one in which they turn into gametocytes.
By inducing overexpression of ap2-g at various times up to 12 h post-RBC invasion, they discovered that gametocytes were produced in that particular RBC cycle whilst, if induction occurred after 12 h, sexual stages were not produced until the next RBC cycle. This rodent malaria thus appears to differ from P. falciparum in respect to timing of sensitivity to the induction of gametocyte commitment. The authors suggest this may reflect the much longer time it takes for P. falciparum gametocytes to develop or that assays to detect a few parasites that may be able to switch to a sexual cycle early in a cycle have not been developed yet.
These experiments also enabled them to conclude that the ratio of male to female gametocytes produced following overexpression of ap2-g remained similar to the parent population.
Transcriptome analysis
The ability to synchronously induce commitment to gametocyte production enabled these colleagues to compare the transcriptome of populations overexpressing or not expressing ap2-g when developing in a mouse host.
The dynamics of the former closely resembled that expected of wild type gametocytes. By 6h post- induction of ap2-g, 56 genes were responding, including many known to be specific to male or female mature gametocytes. These genes had promotor regions enriched in motifs like the DNA-binding motifs of the AP2 region of AP2-G, suggesting they may be downstream targets for AP2-G.
The authors point out that most of these genes are known to be upregulated early in the development of P. falciparum gametocytes. However, in the human malaria parasite, there are genes upregulated and co-expressed with ap2-g that encode proteins not present in the P. berghei genome. These are secreted proteins that are considered to be involved in changing the RBCs that host the parasite, a phenomenon only associated with P. falciparum infection.
The transcriptome analysis also revealed the many of the early response genes had a sex-specific profile, suggesting commitment to one sex or the other precedes, or occurs simultaneously with, AP2-G -mediated gametogenesis.
The next step
It’s known that the initiation of gametocyte production in vivo is flexible and influenced by various environmental factors. However, how differing conditions act upon transcription factors that initiate this developmental pathway is largely unknown. The platform for investigation provided by the overexpression of ap2-g should enable further investigations of the induction and process of gametogenesis in this and related parasites.

Comments