Plasmodium, the parasite that causes malaria, has a famously complicated lifecycle. To be transmitted from human to human, it must cease asexual replication in the erythrocytes of the human host and commit to formation of gametes necessary for sexual replication in the gut of the mosquito vector. Key to this commitment is the differentiation of some of the parasites in the human bloodstream into sexual forms that can mate inside the mosquito. These gametocytes are a dead-end in the human, yet they are essential for transmission: when taken up in a mosquito blood meal, they complete their differentiation into mature gametes and fuse to form a zygote. Asexual forms, conversely, are a dead-end in the mosquito. Thus, the balance between the asexual, replicating forms that sustain the human infection and the sexual, non-replicating forms poised for the bite of a mosquito is essential for efficient transmission of malaria (see figure below). So how does Plasmodium, circulating in the human bloodstream, decide to make gametocytes?
Physiological factors in the environment that trigger sexual commitment are poorly understood, although conditions of stress resulting from a high parasite burden and/or drug therapy are probably involved. The molecular basis of the developmental switch – the “Holy Grail” for understanding gametocytogenesis – has likewise eluded the malaria research community for years. Now, two back-to-back reports in Nature, authored by the teams of Oliver Billker (Wellcome Trust Sanger Institute), Manual Llinás (Princeton and Penn State Universities) and Andy Waters (Glasgow University) have provided a breakthrough. They offer resounding evidence that a conserved member of the Apicomplexan AP2 family of transcriptional factors acts as the master switch in triggering the transcriptional cascade that initiates gametocytogenesis.
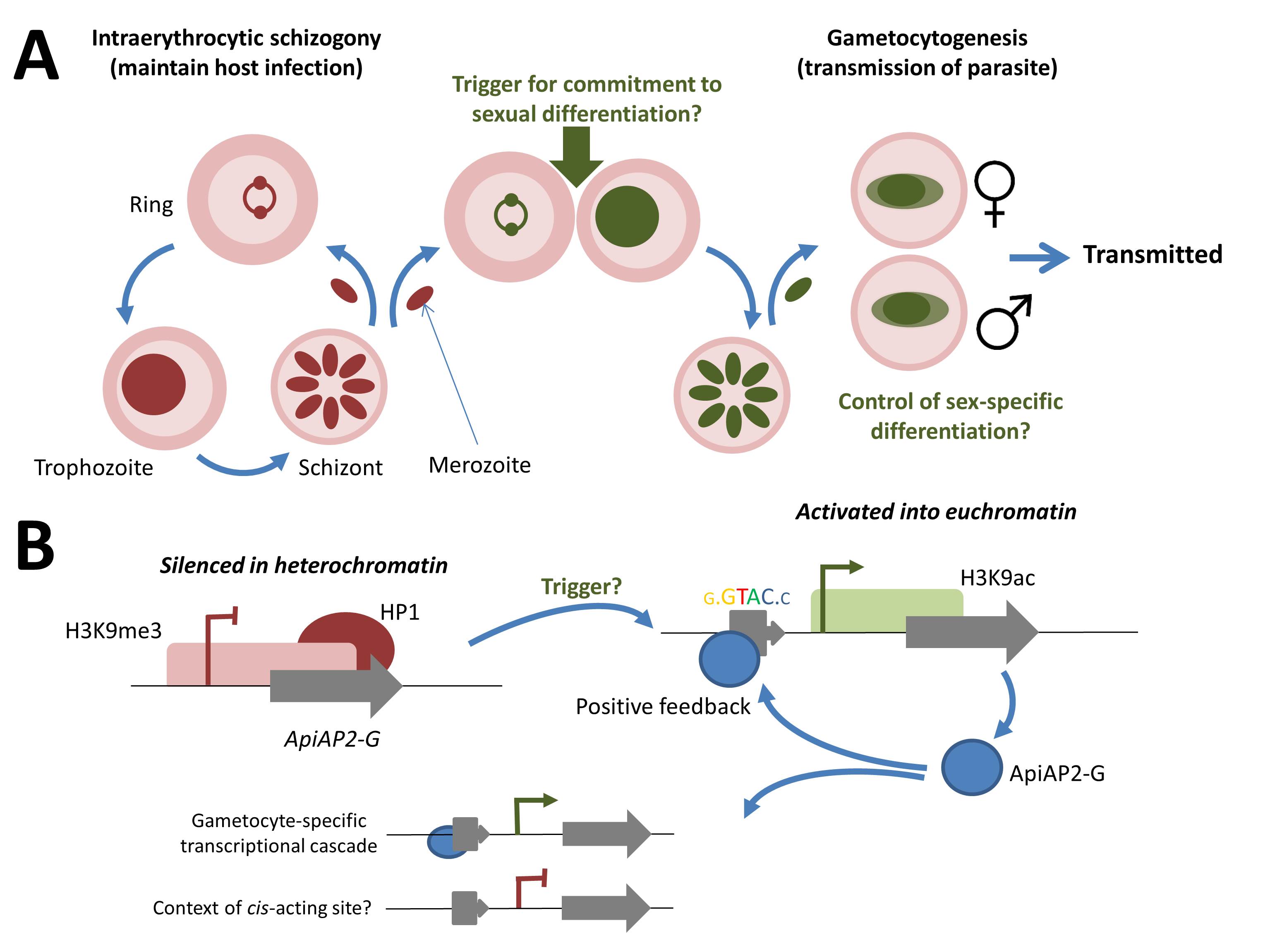
That an AP2 transcription factor provides such a master switch is perhaps not surprising, since they also control developmental transitions during ookinete, oocyst and liver-stage development. What is distinct here is that differentiation in the mosquito and in the liver are immediately necessary for lifecycle progression; commitment to sexual differentiation during the asexual blood stage is not. In fact, it is only required of a small subpopulation of intraerythrocytic parasites, since a low rate of switching provides the necessary gametocytes for onward transmission to a new host.
In these two papers the discovery of the AP2 master switch, termed AP2-G (for gametocytogenesis), is described in two species of Plasmodium. Whole genome sequencing of gametocyte-producing and non-producing isogenic clones in both P. falciparum (human malaria) and P. berghei (mouse malaria) revealed loss-of-function mutations in AP2-G in non-producing clones. By contrast, other genes and regions previously linked to gametocytogenesis showed no such association. Both studies then used forward and reverse genetics to confirm the role of AP2-G in triggering the gametogenic transcriptional cascade. In P. berghei, a second AP2 transcription factor, termed PbAP2-G2, was also identified as necessary for gametocytogenesis, although apparently not acting in a master switch role.
The reports both conclude with a shared hypothesis for the role of AP2-G in the stochastic transcriptional switch that triggers gametocytogenesis. In bloodstream parasites, AP2-G is usually silenced by incorporation into heterochromatin. Indeed, the nucleosomes within the promoter and 5’ region of the open reading frame are decorated with the canonical mark of heterochromatin, H3K9me3, and associated with heterochromatin protein 1 (HP1). Moreover, a pair of insulator-like elements either side of the AP2-G locus in P. falciparum may link the activation/repression of this master switch with the epigenetic mechanisms that govern var gene switching: where mutually-exclusive switching between members of this major family of virulence genes in P. falciparum promotes chronic asexual stage infection via clonal antigenic variation.
The authors propose that an unknown environmental trigger acts to release the AP2-G locus into a euchromatic state (see figure below), so the transcription factor is expressed in all daughter merozoites. These then invade new erythrocytes and are committed to gametocyte formation. Significantly, AP2-G binds to its own promoter as well as to the promoters of downstream effector genes, providing a positive-feedback loop to reinforce its own expression.
These two reports provide a significant step-change in our understanding of the molecular mechanism(s) governing gametocytogenesis and, by extension, transmission of the parasite into the mosquito vector – indeed, the veritable “Holy Grail”. Attention will surely now turn to the control of downstream events and additional effectors such as AP2-G2. For the future, however, gametocytogenesis still harbours many more mysteries. For example, all daughter merozoites from a single cell form the same sex of gametocyte, but the basis of this sex-specific differentiation is unknown. Even more important, if we are to translate the impact of this work is the enigma of the environmental signal for gametocytogenesis. We may now have identified the molecular switch, but what throws that switch, releasing AP2-G from its silenced state and making some malaria patients into ‘transmitters’ while others are not?
Cellular and molecular events during commitment to gametocytogenesis. (A) Replicating asexual blood-stage parasites undergo schizogony, maintaining a chronic infection of the original host. During schizogony an unknown environmental factor (or factors) triggers commitment of all resultant merozoites to gametocytogenesis on reinvasion of the blood cell. The resulting macro- (female) and micro- (male) gametocytes are taken up in a hungry mosquito’s blood meal, completing sexual differentiation and formation of the zygote in the midgut. Following meiosis, returning the parasite to the haploid state, the parasite is reintroduced into a human host (as a sporozoite) during a subsequent blood meal. (B) In the replicative asexual blood stage, the AP2-G locus is maintained in a silenced heterochromatic state. Release from this state, through the unknown trigger, into a euchromatic state provides for expression of the AP2-G protein. Positive feedback is provided by binding to the cognate cis-acting regulatory element located upstream of the AP2-G gene. AP2-G transcription factor binding upstream of genes expressed during the gametocyte cascade providing for downstream effects. Of note, is that the predicted cis-acting motif is not exclusively found upstream of gametocyte-specific genes, suggesting that some additional context is required for this cis-trans transcriptional control.

5 Comments