
The topic of this blog coincides nicely with Charles Darwin’s birthday week. The techniques that we use to study evolution have changed drastically since Darwin’s time, but we still ask many of the same questions.
This week, we’ll be discussing genetics and evolution in one of the dominant malaria mosquito vectors of malaria. Mosquitoes are often cited as the world’s deadliest animal. This is because mosquitoes can carry various types of diseases, including yellow fever, dengue, Zika, and malaria. One genera of mosquitoes, Anopheles, are responsible for 445000 human deaths from malaria each year.
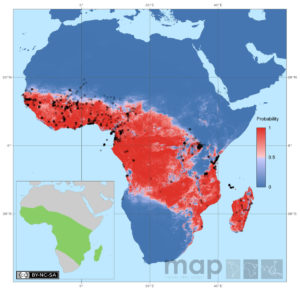
In most regions where malaria is endemic, there is a dominant species of Anopheles that carries malaria. In Africa, where most cases of malaria occur, one of the main mosquito species that is Anopheles gambiae (see left and below).
Understanding mosquitoes is an essential component of the control of malaria. Although treating symptomatic people is a part of malaria control programmes, vector control is an essential component. Mosquito populations are often targeted through bed nets and indoor residual spraying. Bed nets can prevent individuals from getting bitten provided they sleep under the bed nets each night and mosquitoes are feeding then. However, they can also target the populations of mosquitoes when treated with insecticides.
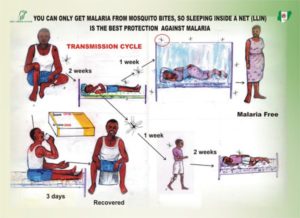
Vector control has become more widespread in recent years in the battle against malaria. This has led to constant to declining malaria prevalence and geographic range. However, in last year’s annual World Malaria Report by the World Health Organization, for the first time in many years, malaria cases in some regions have increased. The thought is that insecticide resistance in mosquitoes is driving this increase. Insecticide resistance has been found in mosquitoes in 60 of 91 malaria endemic countries. This underscores the importance of understanding the diversity and ecology of mosquitoes.
A few months ago, the 10000 Anopheles Genomes Consortium published a paper that aims to better understand genetics of Anopheles. The projects overarching goal is to understand genetic diversity across many species of dominant malaria vectors. This first paper describes genetics of 765 Anopheles gambiae samples from a variety of habitats types (woodland, savanna, rivers) across 15 locations. By sequencing whole genomes of a total of 765 mosquitoes, they were able to ask many questions relevant to mosquito biology and important for mosquito control.
One of the first things the scientist looked at was how much genetic diversity was present within the species. Mosquitoes have one of the highest levels of genetic diversity amongst eukaryotic organisms, and the results of this extensive survey supported that. Within these 765 mosquitoes, the authors identified over 50 million variations within and between populations in the accessible genome. A high level of genetic diversity is often beneficial to a species- it can help a population recover after rapid declines and provides more opportunities for adaptation when a species’ environment changes.
Previous authors have identified some of the genes that can lead to an individual mosquito to be resistant to insecticides. In this study, the authors found evidence of positive selection in recent history, and several genes under selection play a role in insecticide resistance. Not only did they find evidence for insecticide resistance in some individuals, but they also were able to determine that these adaptations have spread through populations, identifying new locations where mosquitoes have insecticide resistance.
The other useful information that one can infer from from genomes is population structure. By comparing populations across Africa, authors were able to infer how much genetic exchange (or successful mating) happens between regions. Surprisingly, North of the Congo basin, mosquito populations are fairly well connected, meaning there is genetic exchange. This has implications for spread of insecticide resistance: if resistance develops to one insecticide in a single location, it may spread to other locations.
This is the first publication to come out of this project and undoubtedly there will be more. Understanding how genetically diverse wild populations of mosquitoes are, how insecticide resistance arises, how it can spread and how likely it is to undergo positive selection will be essential for maintaining current progress in malaria control and pushing towards elimination.

Comments