
The life history strategy known as parasitoidism occurs when an organism lays its eggs in a host and the parasitoid larvae feed on the host, eventually killing it. Many parasitoids are found within the wasp-waisted Apocrita, and use caterpillar larvae as their hosts. During oviposition, some are known to also inject microorganisms that alter the physiology and immune system of their hosts and thereby help maintain the parasitoid’s development. However, they are still under threat because hyperparasitoids parasitise the parasitoids, laying their eggs in parasitoid larvae that are developing concealed inside a caterpillar.
A parasitoid wasp must find its caterpillar host, but hyperparasitoids have a more difficult task as they must recognise which caterpillars are harbouring parasitoids.
How do hyperparasitoids locate their parasitoid hosts?
It has been discovered that detecting odours given off by infected caterpillars may be the answer.
Research published in 2014 demonstrated that caterpillars of the small cabbage white butterfly, Pieris rapae, that were infected with parasitoids, gave off odours that were more attractive to the hyperparasitoid wasp, Baryscapus galactopus, than odours from unparasitised caterpillars.
Where do these odours originate?
Knowing that the microbiome of caterpillars of the related large cabbage white butterfly, Pieris brassicae, is altered when parasitised, collaborators based in the Netherlands and Belgium hypothesised that odour differences detected and preferred by B. galactopus were the result of changes in microorganism-derived volatile organic compounds (mVOCs) given off by infected hosts. The group’s research findings that this was so, were recently published in PLoS Pathogens.
Caterpillar odours
Initially the researchers compared the odours given off by caterpillars of the large cabbage white with those infected with the parasitoid Cotesia glomerata. To take account of the effect of mVOCs given off by the transient internal (gut) and external (skin) microbiome, treatment groups included caterpillars with the gut biome removed by starvation, and groups that were starved and washed to also remove the skin biome.
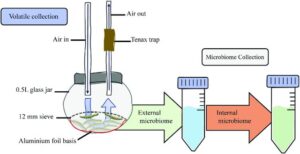
A total of 33 volatiles were identified, with 5 signature VOCs associated with parasitoidism. Most of these had previously been identified as alarm pheromones in other insects. Although starvation and removal of the surface microbiome affected the volatile profile, differences were still found between treated parasitised and unparasitised caterpillars, suggesting microorganisms passing through the gut, or present on the skin, were not involved in signalling the presence of parasitoids.
The source of mVOCs
DNA was initially collected from the caterpillar’s surface, and then from dissected caterpillars, and 16S ribosomal RNA (rRNA) gene sequences examined. This revealed a considerable difference between the external and internal microbiome. The presence of parasitoids significantly affected both internal and external microbiome, with the diversity of the external microbiome decreasing and the internal microbiome increasing. Interestingly, one microbe, a Wolbachia strain, dominated the internal microbiome of parasitised caterpillars, but was not present on the surface. This bacterium was not found in the microbiome of uninfected caterpillars. The authors suggest B. galactopus females inject Wolbachia into the caterpillar during egg laying.

The hyperparasitoid’s odour choice
The investigators then looked at the hyperparasitoid’s response to the smell of differently treated caterpillars. They used a Y-tube olfactometer to present 1000 B. galactopus females a choice between odours from different treatment groups in turn. Over 84% responded by moving down one or other tube. After removal of the gut microbiome by starvation they did not distinguish between odours from parasitised and nonparasitised caterpillars in this assay. However, significantly fewer hyperparasitoids chose odour from starved plus washed parasitised caterpillars compared with starvation alone. Although disruption of the external microbiome caused a loss of preference, there was no such difference in preference when these treatments were performed on uninfected caterpillars. It would appear that the parasitoid’s presence caused changes in mVOCs from the gut and external microbiome that the hyperparasites could detect.
Is Wolbachia involved in attracting the hyperparasitoid?
This question was tested by measuring the time it took B. galactopus wasps to land on caterpillars injected with Wolbachia, compared with parasitised and nonparasitised caterpillars. The hyperparasitoids landed significantly more times on parasitised than nonparasitised caterpillars and also more on Wolbachia-injected than non-treated ones. However, once they landed, more time was spent on parasitised hosts than Wolbachia-injected or non-treated ones. This suggested additional clues, not associated with the bacterium, are involved in the identification of the presence of a parasitoid once initial attraction has occurred.
The potential role of Wolbachia is this complex association of insects is particularly interesting. This inter cellular symbiont is known to affect the metabolism, immune and reproductive systems of many insects. It is conceivable that volatile chemicals produce by, or induced by its presence, could diffuse out of the caterpillar spiracles, signalling the presence of a parasitoid larvae hidden inside.
Conclusions
The authors make the following conclusions:
- Infection with the parasitoid changes the odour of cabbage white butterfly caterpillars and these changes are associated with changes in its bacterial composition.
- The hyperparasitoid preferred odours from starved caterpillars over those from caterpillars that had also had the external microbiome removed.
- Wolbachiabacteria are only present in parasitised caterpillars and their presence attracts the hyperparasitoid wasp, but the mechanism involved is, so far, unclear.
They acknowledge differences in their findings compared with previous work on this system and point to an important role for environmental factors, such as caterpillar diet and temperature, on odour production that may influence findings. The authors identify the need to characterise the mVOCs produced by Wolbachia and test them in behavioural assays.
In addition, they point out that a clearer understanding of the biology of hyperparasitoidism has commercial implications because parasitoids are used in the biological control of horticultural pests, particularly in green houses. The presence of hyperparasitoids that are killing these useful assassins could be avoided by employing specific odour-baited traps.

Comments