Guest post by James Edwards-Smallbone, Keele University, UK
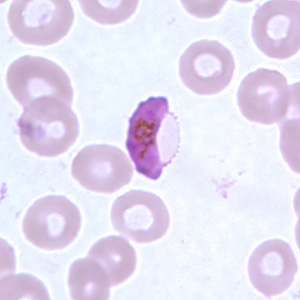
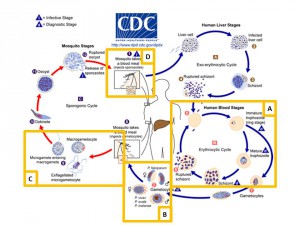
The malaria parasite, Plasmodium falciparum, is responsible for about 200 million infections per year which impose a high burden of morbidity in endemic countries and is the cause of at least half a million deaths every year, mostly in children under the age of five. Following invasion and egress from hepatocytes the parasite undergoes multiple rounds of invasion, replication, host cell lysis, and reinvasion in red blood cells (Figure 1, Box A). However, in response to host cues such as red cell depletion and immunity, and a high parasite burden Plasmodium can commit to a developmental pathway that leads to differentiation into either male or female gametocytes (Figure 1, Box B, Figure 2). A strong candidate for the developmental trigger that controls this is a member of the AP2 transcription factor family as detailed in a previous post.
Gametocytes are essential for transmission to the vector (a species of mosquito in the genus Anopheles) and in turn parasites are able to spread to subsequent human hosts (Figure 1, Box D). Sexual reproduction in the mosquito (Figure 1, Box C) is also an important source of genetic recombination, generating new parasite variants that enable long term persistence in the host population. Gametocyte formation is thus a key part of the Plasmodium lifecycle and a promising source of targets for transmission-blocking drugs and vaccines. However, due to the low conversion rate of blood-stage parasites into gametocytes and the fact that gametocyte-positive patients may be asymptomatic it is frequently difficult to identify infectious individuals. There is thus a pressing need to increase the availability of specific diagnostic markers.
In addition, partitioning into male and female gametocytes is a relatively poorly-explored process. There is evidence to suggest that the ratio of male to female gametocytes is variable in the host and that this may influence human to mosquito transmission. As such, an improved overview of the partitioning process may yield insights into transmission dynamics between humans and mosquitoes.
A new study by Tao and colleagues aims to improve understanding of sex partitioning in P. falciparum gametocytes and uncover sex-specific markers using a proteomics approach. The researchers combined analysis of mass spectrometry data gathered from two strains of P. falciparum with a reanalysis of data previously acquired for the rodent malaria, P. berghei, in an earlier study by Khan and co-workers.
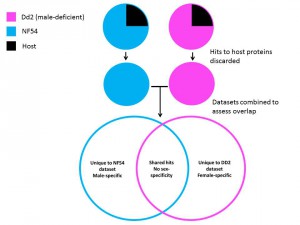
The two strains used by the group in their study informed their approach as one (Dd2) is deficient in formation of stage V (the final developmental stage) male gametocytes. Unique protein identifications from this strain could therefore be ascribed to female-only markers, and likewise unique hits to the other strain (NF54) could be defined as male-specific, once hits to host proteins had been discounted (Figure 2).
Protein hits from mass spectrometry data are ascribed identities by searching peptide fragments against a protein database. Therefore the more extensive and up-to-date this database is, the more reliable any protein identifications that are made will be.
The researchers wished to interpret their findings from P. falciparum based on previous data acquired from P. berghei by Khan et al. in 2005. To account for improvements in gene annotation made since that study they re-analyzed that same dataset, noting a general increase in the number of proteins identified for male-specific, female-specific and non-specific proteins, with 40-50% of the original identities conserved into the new analysis. A small number of proteins were re-assigned from male to female and vice versa compared with the original analysis, but these represented only a small fraction of the total number of identified proteins in each case.
Interestingly Tao and colleagues were also able to identify two male-specific promotor genes, detailed in reports unrelated to the Khan study, which segregated as male. Conversely the researchers were also able to confirm a separate report that one gene (Male-development gene 1), previously linked to male gametocytogenesis, did not partition as male on re-analysis.
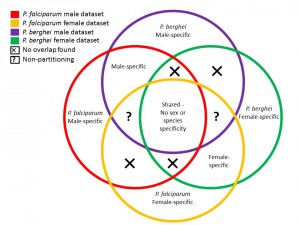
Having two datasets for sex-partitioning of P. falciparum (NF54/Dd2) and P.berghei (Khan et al. dataset) the researches undertook a series of comparisons between the datasets (systematic subtractive bioinformatics) to systematically isolate unique protein groups (illustrated schematically in Figure 3). From these analyses they were able to identify a large pool of proteins of unknown function which partitioned either as male or female from either species.
Do the proteins from these partitioned lists have biological relevance? Tao and colleagues investigated this question by selecting three putative markers for female gametocytes from the partitioned protein sets and generating antibodies against them. They then tested these antibodies using Western blot and immunofluorescence assays, discovering that two of the three markers displayed specificity for female gametocytes.
Future efforts will focus on verifying and characterising these two genes as potential diagnostic markers or targets for transmission-blocking interventions. Other sex-specific proteins from the partitioned pools will also provide researchers in the field with a valuable resource and it is likely that other candidates for diagnostic or therapeutic purposes may emerge. The Plasmodium research community now has a powerful new means to answer fundamental questions about gametocyte biology and development. This can only improve prospects for individuals and countries afflicted with malaria.

Comments