
Onchocerciasis
Onchocerciasis, or “river blindness” is a parasitic disease caused by the filarial worm Onchocerca volvulus. Over 100 million people are at risk of infection, of which 99% reside in 31 sub-Saharan African countries endemic for onchocerciasis.
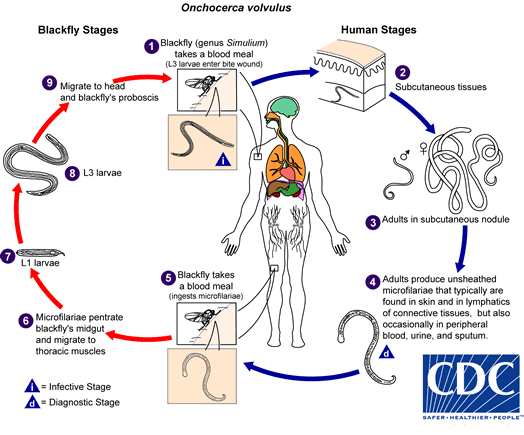
Onchocerciasis is transmitted by an infected blackfly (genus Simulium) during a blood meal. Once in the subcutaneous tissues the parasite larvae develop into adult filariae, which commonly reside in vascularized nodules in subcutaneous connective tissues. Adult worms can live in nodules for up to 10–15 years. The female worms mate and produce larvae called microfilariae. Microfilariae live for approximately 12–15 months in the skin and can also migrate to the eye. A blackfly ingests the microfilariae during a blood meal and the microfilariae successively develop into third-stage infective larvae within the vector. The blackfly bite can then infect a human with third-stage infective larvae.
There are a variety of symptoms associated with the disease and most symptoms are a result of immune response to dead or dying microfilariae. Inflammation in the skin causes intense itching and can result in long-term damage, such as changes in skin color, texture and elasticity. Inflammation may also occur in the eyes as a result of microfilarial death, which can result in blindness if left untreated.
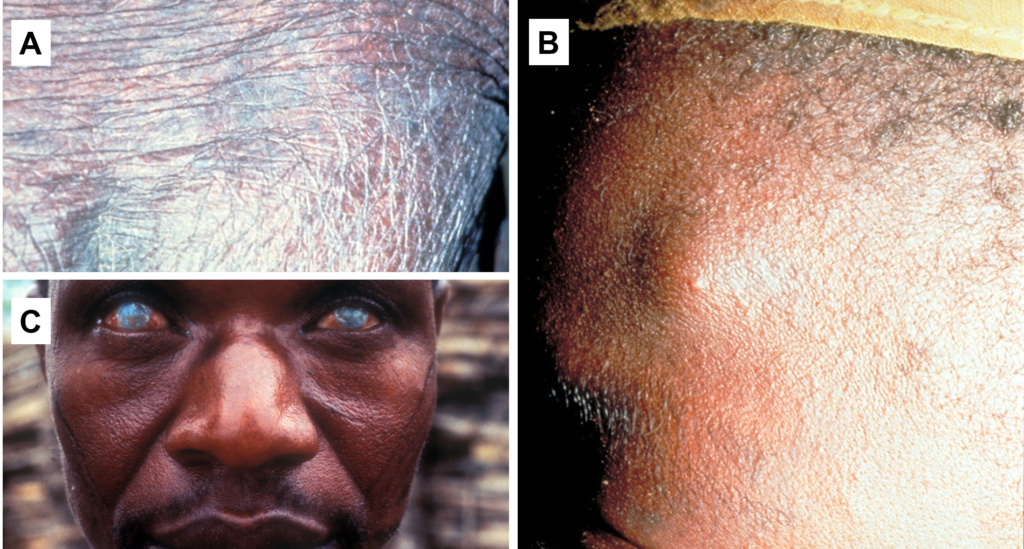
Diagnosis of onchocerciasis
In Africa, the standard elimination strategy is annual mass drug administration (MDA) with ivermectin. Ivermectin kills microfilariae in the skin but does not kill the adult worms in skin nodules.

World Health Organization (WHO) guidelines for stopping MDA and verifying onchocerciasis elimination recommend serological evaluation in sentinel populations by anti-Ov16 ELISA. However, serological testing cannot distinguish between historical and current infection.
Skin samples or ‘skin snips’ can also be analyzed for microfilariae using microscopy or molecular techniques. This is not recommended by the WHO as skin snip analysis has reduced sensitivity when microfilarial numbers are low or absent, such as after treatment with ivermectin. Skin snips are also invasive and becoming increasingly unpopular.
A specific and sensitive diagnostic test is therefore needed for onchocerciasis ‘end-game’ scenarios, both to verify elimination and to detect cases when endemicity levels no longer justify MDA. A potential alternative to skin snipping could be detection of parasite nucleic acids in the host’s blood. Onchocercomas, the subcutaneous nodules containing adult worms, are highly vascularized, and so O. volvulus DNA or microRNAs (miRNA) may be detectable in blood. This method would also not be reliant on the temporal presence of microfilaridermia for diagnosis.
In light of this, we conducted a study to assess the potential of circulating O. volvulus nucleic acids to diagnose infection and track antifilarial treatment efficacy.
The study
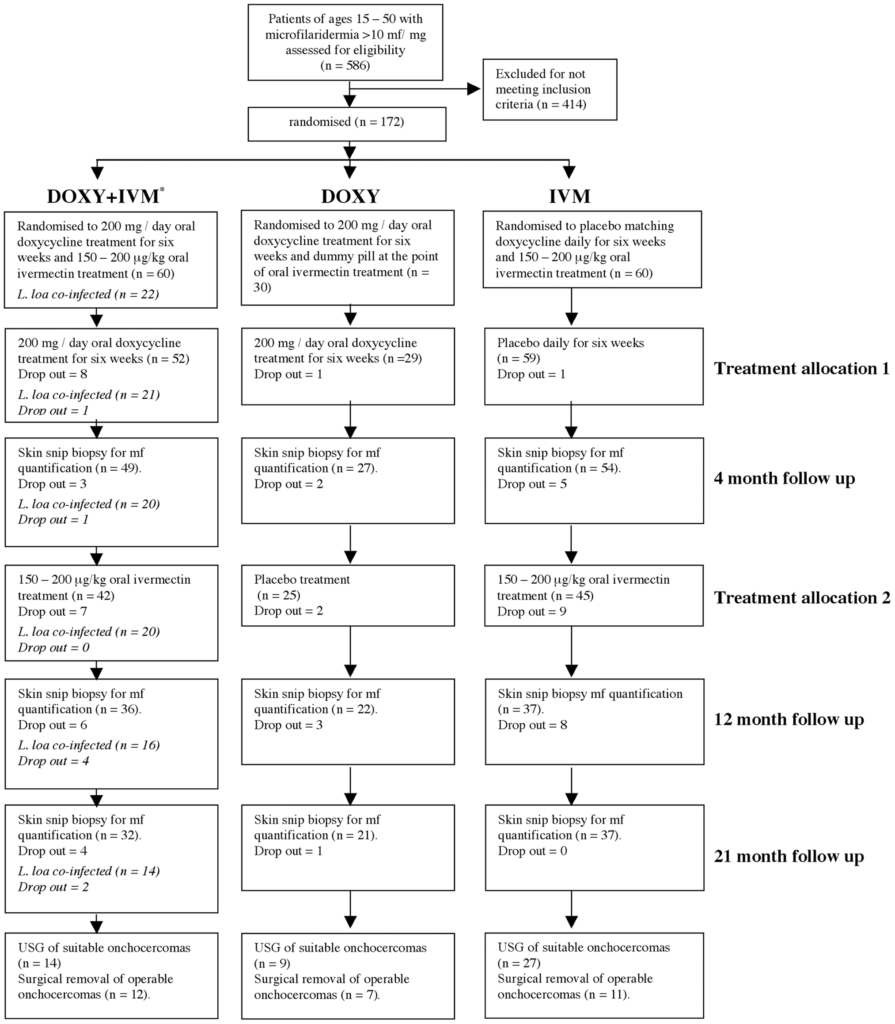
A longitudinal plasma sample set was previously collected over 2003-2005 in Cameroon during a randomised trial with onchocerciasis-infected individuals.
Patients had skin snips and plasma collected at baseline and then at four, 12 and 21 months after receiving either microfilaricidal (ivermectin), macrofilaricidal (doxycycline) or combination treatment. In the current diagnostics study, participants with co-infections were omitted and the sample-set included: doxycycline (n = 9), doxycycline + ivermectin (n = 5) and ivermectin (n = 4).
RT-qPCR was used to detect parasite-derived miRNAs lin-4 and miR-71 (previously sequenced in plasma from O. volvulus-infected individuals in Cameroon) and qPCR was used to detect the O. volvulus-specific O-150 repeat DNA sequence.
Additional novel O. volvulus DNA repeat families identified through bioinformatic approaches were also investigated as alternative targets to the O-150 repeat DNA.
Circulating nucleic acids for detection of onchocerciasis
After removing samples with known parasite co-infections, 18 patients (72 plasma samples) were investigated across the three treatment groups. Parasite-derived miRNAs were very weakly detected in two of 72 plasma samples (2.8%) and two of 47 samples (4.3%) with microfilaridermia using RT-qPCR. miR-71 was detected in a doxycycline-treated patient at month four and lin-4 was detected in a doxycycline-treated patient at month 12.
The O-150 DNA sequence was detected in eight (44.4%) baseline plasma samples by qPCR and the number of positive samples declined after treatment. Two individuals were positive at both baseline and month four but negative at later time points, and one individual remained O-150 positive over 21 months. Of the 72 samples evaluated, 47 were microfilaridermic and 14 were O-150 positive. Among the samples with microfilariae present in skin snips, only 11 (23.4%) were O-150 positive.
After conducting bioinformatic analyses, 10 novel DNA repeat families were selected for further evaluation. While the O-150 repeat region was predicted to occur 46 times, several repeat families were predicted to occur >1,000 times. However, qPCR analysis revealed the novel DNA repeat families were comparatively less abundant than the O-150 repeat.
Conclusions
Our research has demonstrated that O. volvulus nucleic acids are variably detectable at low to undetectable concentrations in host plasma. While the O-150 DNA target showed the greatest frequency, qPCR testing of plasma was well below the sensitivity of parasitological diagnosis. Despite encouraging bioinformatic predictions, all novel DNA targets were less abundant compared to O-150. Circulating parasite-derived nucleic acids are therefore insufficient as diagnostic tools or as biomarkers of treatment efficacy for onchocerciasis.

Comments