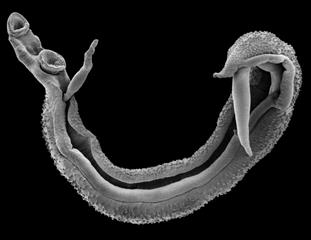
The parasite
Schistosoma mansoni, the causative agent of intestinal schistosomiasis, is a nasty yet intriguing little parasite. Schistosomes predominantly live in pairs in the blood system of mammals, pumping out eggs that have to somehow make their way out into the big, bad world (or nearest water body) in order to infect a snail, clonally multiply and emerge in the water to locate their own little mammalian niche. However, a lot of the eggs (though we don’t know what proportion exactly) don’t make it out, being swept by the flow of blood into neighbouring organs, lodging in the tissue and dying. This is what leads to the most damaging outcome of the disease, as the host’s immune system detects the foreign object (the dying egg) and goes into attack overdrive, causing tissue damage to build up in the organs.
Whilst there is a treatment for schistosome infections, called Praziquantel, there are two major issues with it:
- It does not prevent re-infection, meaning treatment has to be repeated regularly; called preventative chemotherapy (PC).
- It isn’t 100% effective against S. mansoni.
Scientists and public health experts would love to have an effective vaccine against schistosome infections. This could be a game-changer in the fight against schistosomiasis. However there are numerous challenges hindering this goal. One of them is the lack of an appropriate cost-effective model to test a vaccine candidate.
Purpose of the study
This study aims to assess the safety of developing a controlled human infection model (CHIM) for testing treatment interventions. If safe this will allow testing of the effectiveness of potential schistosomiasis vaccines.
Whilst there are a few candidates for schistosomiasis vaccines, carrying out vaccine field-trials can be prohibitively expensive. In order to secure the funding, scientists have to show convincing preliminary evidence that the vaccine candidate works and is safe for both schistosome naïve individuals and for people who have had a history of schistosomiasis. The latter is currently being tested for one of the vaccine candidates: the Sm-TSP-2 antigen is being trialled in an endemic area of Brazil by investigators from the George Washington University and the René Rachou Institute of the Oswaldo Cruz Foundation (FIOCRUZ) of the federal Brazilian government. If this clinical trial proves successful, the next step is to show that it does work as a vaccine against schistosomes in humans. This is where this controlled human infection model (CHIM) could prove very useful.
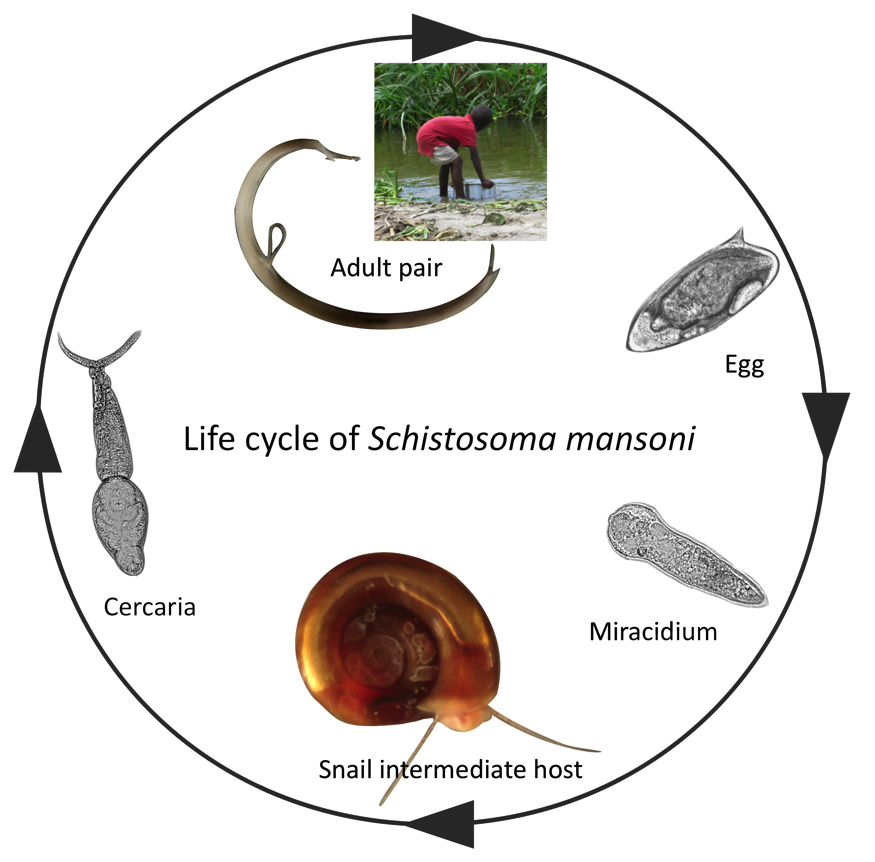
The process
Selecting volunteers followed a long list of inclusion and exclusion criteria to mitigate health risks, and confounders.
Inclusion Criteria:
- Aged between ≥ 18 and ≤ 45 years and in good health.
- Adequate understanding of the procedures of the study.
- Good communication with the investigator and will commit to all the study visits.
- Will remain within Europe (excluding Corsica, since there could be schistosomiasis transmission there, check out our blog on this) during the study period and reachable by mobile telephone from week 3 to week 12 of the study period.
- Agrees to refrain from blood donation throughout the study period.
- Women in particular must agree to use adequate contraception and not to breastfeed for the duration of study.
- Has signed informed consent.
Exclusion Criteria:
- Anyone with a history, or evidence at screening, of a whole range of diseases and symptoms from cardiovascular to psychiatric. Including HIV, hepatitis B or C, history of drug or alcohol abuse, malignancy of organs etc.
- Anyone using any drug known to interact with praziquantel.
- Women who test positive for pregnancy.
- Anyone with a history of schistosomiasis or treatment for schistosomiasis.
- Anyone testing positive by serology for schistosomiasis or elevated serum or urine circulating anodic antigen or positive Schistosoma serology at baseline.
- Anyone known to be hypersensitivite to, or contra-indications (including co-medication) for, use of praziquantel or, artesunate or lumefantrine.
- And anyone who is an an employee or student of the department of parasitology or infectious diseases of the Leiden University Medical Center.
Once volunteers who had met all the above criteria were selected, they and their soon-to-be parasitic guests where prepped for infection.
The team used the Puerto Rican lab strain of S. mansoni, that had been passaged through laboratory mice and snails. 20 male-only viable cercariae per volunteer were used. The use of single-sex collections was to avoid pairing schistosomes and the associated health risk of schistosome egg production. These 20 male-only cercariae were placed on the arm of each volunteer in a nice little drop of warm water to increase the likelihood of cercarial penetration. Volunteers reported a slight localised rash, presumably around the area of penetration.
Currently the parasites are assumed to be residing happily in their new host’s blood system, where they are expected to stay for 12 weeks. The volunteers will visit the research centrer every week where a blood sample will be collected and analysed, particularly for CAA (circulating anodic antigen), a molecule excreted by worms, indicating that the worms are still alive and present.
After the12 week period the volunteers will be treated with Praziquantel. Let’s hope it kills all the worms!
The response
Volunteers are being paid for their time but many have expressed personal or professional interest in this experiment. One of the volunteers has a scientific background and is interested in vaccine development and another has come from East Africa and knows first hand the devastating impact the disease can have on people.
This study has certainly captured peoples’ imagination, being widely shared on blogs (including this one), news sites and social media, with outpourings of admiration, dismay and gratefulness from people across the globe.
If successful it could be a very useful tool for the development of new interventions in schistosomiasis prevention
.

Comments