Diagnosis of diffuse gliomas
Diffuse gliomas, including glioblastomas, are the most common type of malignant cancers that arise in the brain or spinal cord of a patient. Depending on the type of glioma that a patient has, standardized treatments are relatively non-specific and can include surgery, radiation, chemotherapy, or a combination of these therapies. Also depending on the type of glioma, there are different predicted average survival times ranging from months to years.
Diagnosis of a glioma (or any brain tumor for that matter) is usually made by a neuropathologist such as myself, using a microscope to examine surgical tissue removed from a patient by biopsy or resection. In addition to microscopic examination of brain tumors, a number of genetic tests are available that can also provide additional information about the relative aggressive behavior that an individual glioma may have. These are limited in number, but provide a powerful addition to diagnosis, and in 2016, the World Health Organisation re-classified diffuse gliomas according to molecular parameters as well as traditional histology. There is a continuous need refine the definitions of gliomas based on their genetic signatures in order to better predict clinical outcome and help guide therapy, which is often referred to under the umbrella of ‘precision medicine’ (for a review, see here).
A new molecular classification scheme
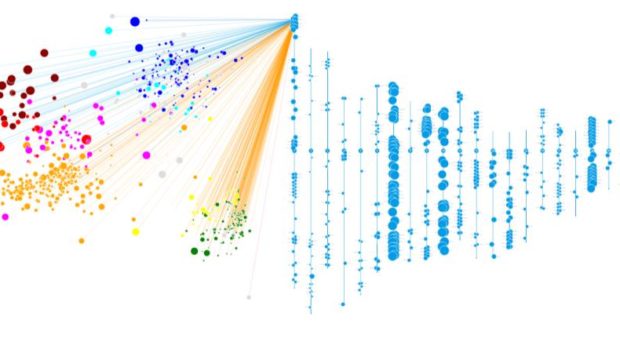
To better understand the genetics of gliomas, Dr. Eric Holland (Senior Vice President and Director of Human Biology at the Fred Hutchinson Cancer Research Center) along with colleague Dr. Hamid Bolouri, developed a visualization technique that represented how closely related gliomas are based on their genetic signature. Their paper about big data visualization for gliomas was published in 2016 and they have launched a web-based, interactive data mining tool termed Oncoscape.
Working in close collaboration with Dr. Holland, we recently used Oncoscape as a platform to visualize gliomas according to the World Health Organization’s (WHO) classification scheme, which is the standard classification by which brain tumors are diagnosed. Importantly, we were able to go beyond standard WHO classifications, and derive a new molecular classification scheme based on relatively few genetic markers that predicted clinical outcome (see Figure 6 for a visual summary). We further validated the prognostic utility of this molecular classification in a large independent cohort of glioblastomas. This classification system has the potential basis to be incorporated into routine clinical practice, with the hopes that it would improve the quality of life of an individual patient with brain cancer through accurate diagnostics and risk-stratification.
Precision medicine for all cancer types
Moving beyond the brain, our work provides a ready-made initial platform to do similar analysis for many other cancer types (breast, lung, gastrointestinal, skin, etc). Refining cancer classifications based upon integrated genetic signatures in a user-friendly visual spatial arena offers the hope of pushing forward precision medicine across all cancer types. For me, this is a truly exciting time to not only be a diagnostician, but a cancer researcher trying to incorporate new technologies and genetic understanding of cancer to improve the lives of glioma patients through a lab bench-to-bedside approach.
Comments