
Insecticide resistance
The recent World malaria report highlights resistance in Anopheles mosquitoes (the major vectors of malaria) to at least one of the four most commonly-used insecticides, across 73 malaria-endemic countries. Furthermore, in 28 of those countries, insecticide resistance has been reported in all main insecticide classes.
This finding is particularly concerning as malaria prevention relies heavily on mosquito control interventions that use these chemical insecticides, whether through residual spraying (IRS) or via use in insecticide-treated bed nets (ITNs).
This emerging resistance of Anopheles populations to insecticides, such as deltamethrin (a common pyrethroid insecticide), has been attributed to their extensive, sustained, and often indiscriminate use across much of Africa.
One important mechanism reportedly involved in mosquito resistance to insecticides is target-site insensitivity, whereby knockdown mutations (kdr) occurring in voltage-gated sodium channels in Anopheles mosquitoes results in resistance to pyrethroid and organochlorine insecticides.
Two mutations in particular, L1014F and L1014S, are frequently detected in Anopheles populations from African countries, and these are used as molecular markers for pyrethroid resistance in Anopheles populations.
Despite surveillance programs to detect instances of insecticide resistance being relatively well-established for most African countries, there are certain countries and areas that have not received as much attention.
One such country is Cabo Verde, a volcanic, tropical archipelago located approximately 450 km off the west coast of Africa, which is home to around half a million people. Of those 500 thousand, around 60% live on Santiago, the largest island within Cabo Verde.
Currently, the extent to which insecticide resistance due to L1014F and L1014S mutations in Anopheles population on Cabo Verde is extremely limited, and thus also poorly understood.
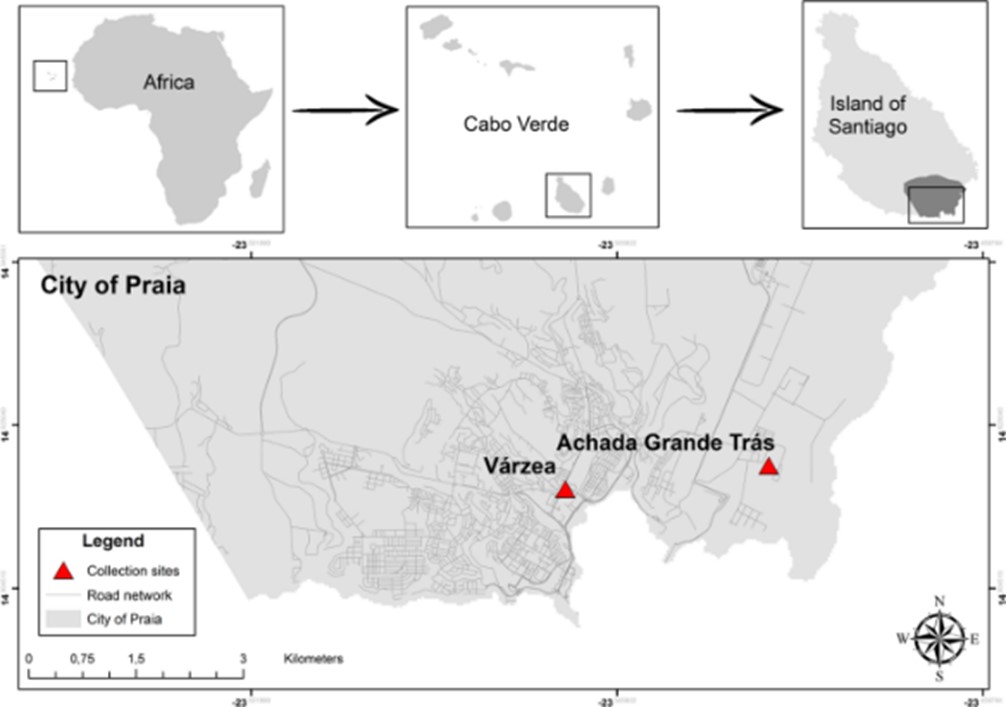
Therefore, a recent study by Derciliano Lopes da Cruz and colleagues from Oswaldo Cruz Foundation in Brazil set out to investigate the prevalence and distribution of resistance alleles L1014F and L1014S in natural Anopheles arabiensis populations from Santiago Island, in Cabo Verde.
Mosquito collection and identification
Initially, mosquito larvae were collected from water sources from two sites in Praia city using a dipper. They were then sent to Jean Piaget University and reared until the adult stage.
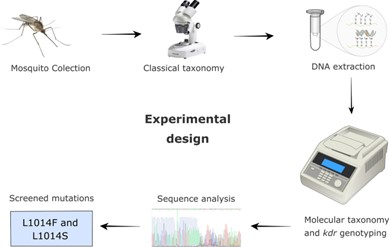
Adult mosquitoes were then identified using microscopy based on morphological characteristics to ensure collected mosquitoes were from the Anopheles genus.
Anopheles mosquitoes specifically from the An. gambiae species complex (of which An. arabiensis is a part) were then selected for molecular identification using PCR to support morphological identification via microscopy.
These mosquitoes were then screened for the L1014F and L1014S kdr mutations, with PCR products sent for Sanger sequencing.
Frequency of insecticide resistance-inferring mutations among collected mosquitoes
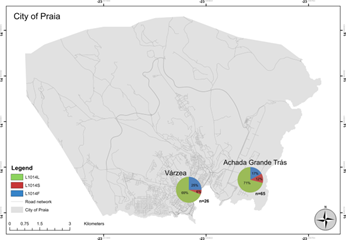
In total, 105 adult Anopheles mosquitoes belonging to the An. gambiae complex were identified using microscopy techniques, and were confirmed to be An. arabiensis using PCR. Of those 105, 91 produced informative gene sequences for down-stream screening for mutations.
Based on the sequences produced, 52 were homozygous for the susceptible genotype (SS), meaning no resistance markers were identified.
For the L1014F mutation, 15 mosquitoes were heterozygous (RS), meaning they contained one allele that inferred resistance, and 10 were homozygous (RR), meaning both alleles contained the resistance-inferring mutation. Similarly for L1014S, 10 were determined to be heterozygous (RS), and four were homozygous (RR).
This results in a frequency of approximately 19% (L1014F) and 10% (L1014S) of the sampled Anopheles populations from Cabo Verde as being resistant to pyrethroid insecticides to some extent. Worryingly, the frequency of L1014S demonstrated here has increased from when it was first identified in 2015, growing from 7.3% to 10%. This finding may indicates that resistance to insecticides is being selected for, and that local mosquito populations are adapting to withstand the local vector control programs. Indeed, the kdr mutations found here may be responsible for resistance to deltamethrin previously established in Anopheles mosquitoes from Praia.
Implications for Anopheles vector control and prevention of malaria in Cabo Verde, and beyond.
The fact that both mutations were found at both study sites suggests that insecticide-resistant mosquitoes may be well distributed across Praia, a dense population centre, where vector control using these chemical insecticides is most common.
Although the frequency of insecticide resistance in Anopheles mosquitoes reported in Cabo Verde is less than has been established for other African countries, these results show, for the first time, that the L1014F resistance allele is present in Cabo Verde Anopheles mosquitoes, and that the frequency of the L1014S mutation is increasing. These findings emphasis the need for continued and improved surveillance of mutations associated with insecticide resistance in Cabo Verde, as well as across the entire continent (and beyond).
In addition to expanding surveillance for insecticide resistant mutations, the authors note that future work should include phenotypic analyses to determine the phenotype of resistance to pyrethroids, and thus ideally the molecular mechanisms underpinning this resistance.

Comments