Advances in microscopy and imaging (e.g. histology, confocal microscopy, X-ray tomography, ultrasounds, and MRI) enhance our understanding of many biological processes. Three-dimensional representations of complex structures obtained with different imaging modalities (e.g. X-ray computed tomography and magnetic resonance imaging) have been especially useful and impressive in vertebrates (including humans).
However, imaging whole live invertebrates (e.g. insects) is very challenging. Apart from the acute radiation doses required to obtain the desired image when X-rays modalities are used, movement of the live subjects compromises image quality by producing artefacts. Therefore, research involving invertebrate imaging is usually destructive and requires animal sacrifice, especially in time-course studies.
To image live insects, it is crucial to fully immobilize the individuals, deliver a sufficiently low radiation dose to allow repeated scans of the same individuals for time-course studies (when X-rays are used), and provide adequate image resolution and quality to distinguish in situ structures. Moreover, the method should have minimal or no subsequent negative effects on life history traits (e.g. growth, reproduction, and longevity) or physiological processes (i.e. normal biological functions) of test subjects.
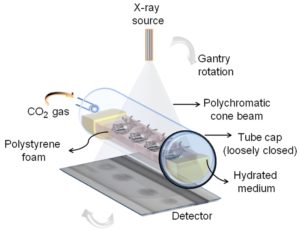
Therefore, we devised a method using carbon dioxide (CO2) gas as anesthetic to induce hypoxia (oxygen deficient conditions) that prevents motion artifacts during non-invasive imaging of whole live insects for 3D visualization. With this approach (summarised in Fig. 1), we were able to keep the live insects immobile for a prolonged period (up to 7 hours), and repeatedly scan the same individuals using X-ray micro-computed tomography (micro-CT). We also demonstrated that if the correct scan protocol is used those individuals recover with minimal impact on their subsequent behaviour and reproduction.
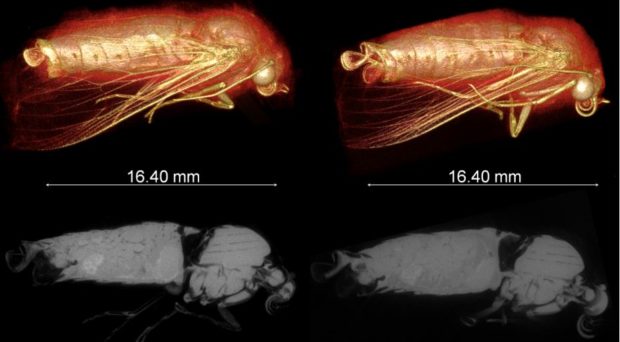
We have overcome the technical challenges of live insect imaging by relying on these organisms’ remarkable abilities to survive prolonged hypoxia and ionising radiation doses (at least 100x more than the lethal dose for humans). We successfully employed CO2 gas as an anesthetic to provide temporary paralysis to keep the insect fully immobilized during micro-CT scanning (similar to a human CT scan where the patient must remain as still as possible) to provide 3D visualization (Figs. 2 & 3).
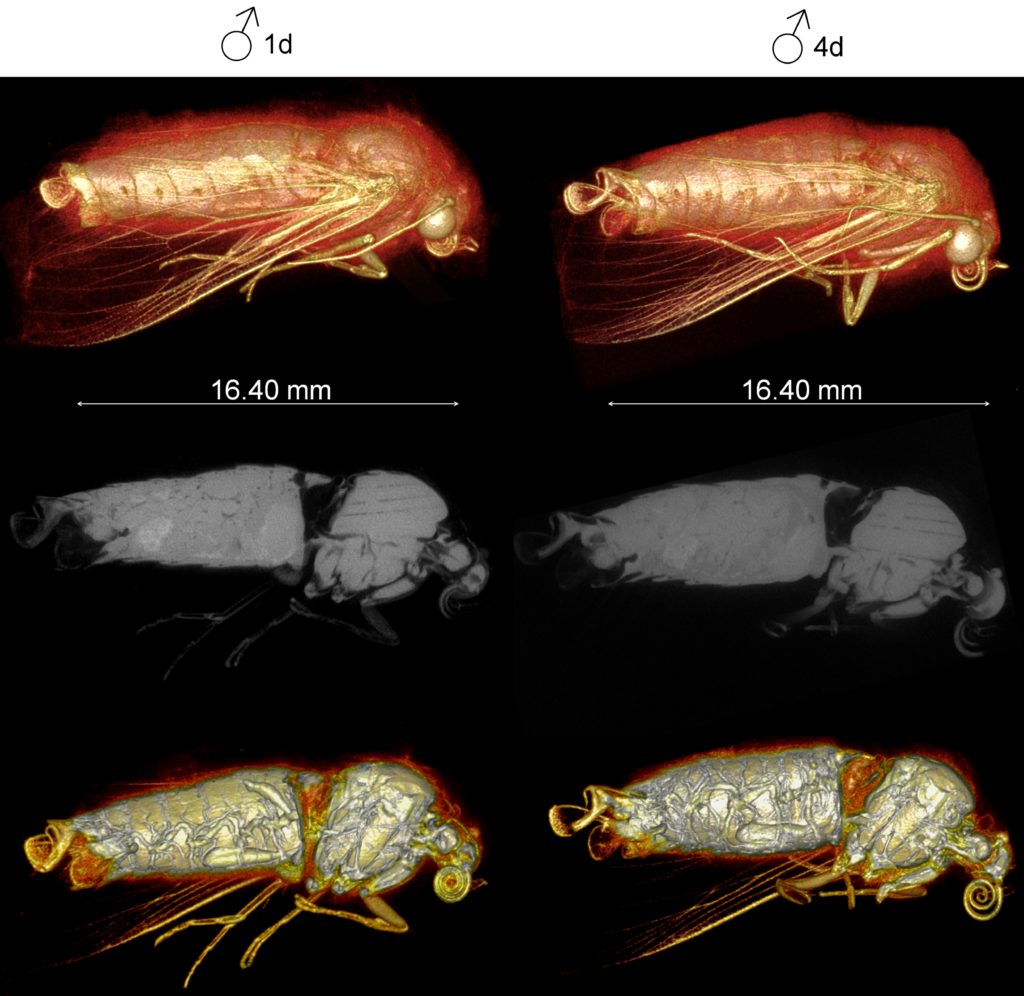
Since we used X-ray micro-CT here, establishing an optimal scanning condition was key due to the unavoidable trade-off in the dose-to-noise relationship (i.e. lower radiation dose = low image quality due to noise, and vice-versa). Our optimised scanning condition produced radiation doses that are at least 80 times less than those used for insect sterilization. Under the optimal condition, insect adults are kept fully immobilized for long enough at relatively low radiation and hypoxic doses from which they can recover rapidly.
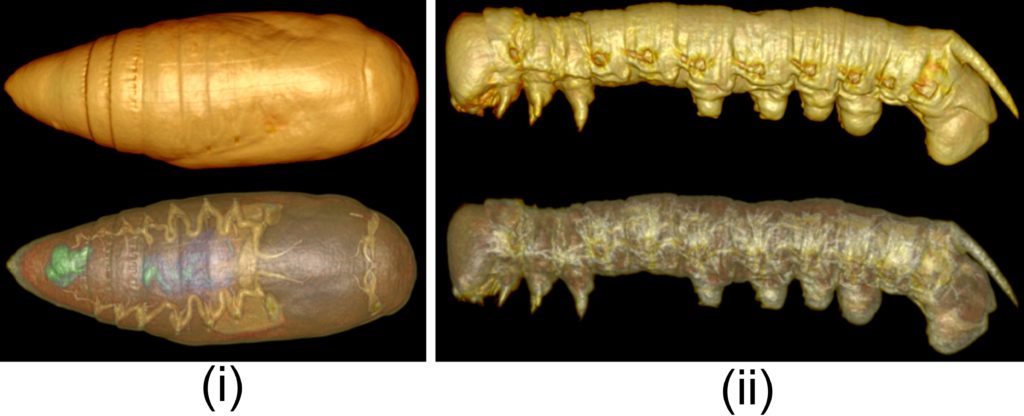
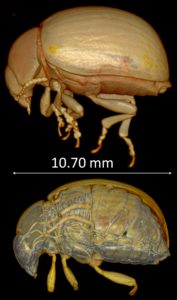
In summary, we successfully exposed live insects (adults and other life stages; see Fig.4) to CO2 gas as an anesthetic for whole-body unrestricted immobilization during X-ray micro-CT imaging, with little or no impact on their recovery and behaviour. We believe this new way to visualize live insects can be easily implemented and is compatible with other imaging modalities (e.g. MRI). Within its limitation, this imaging method has various applications in developmental studies (especially in time-course studies) in a range of disciplines such as medical entomology and parasitology.
Comments