
Hepatitis B – What it is and why it matters
The World Health Organization (WHO) estimates 2 billion individuals have been infected by Hepatitis B virus (HBV) at some point during their lifetime. Within the first 6 months after an individual is first infected with HBV, they experience a short term (acute) illness. In some cases of acute infection, the HBV remains in an individual’s body causing them to be chronically infected with Hepatitis B. The 2015 WHO report on Hepatitis estimated 257 million people (3.5% of the global population) were living with a chronic HBV infection.
Since HBV primarily attacks the liver, chronic HBV infection can imply significant long term health problems including liver damage, failure or even death. Untreated chronic infection can lead to life threatening long term complications such as chronic hepatitis, cirrhosis and hepatocellular carcinoma (a form of liver cancer). HBV is the 10th leading cause of death globally and results in 500,000 to 700,000 deaths annually. Although relatively rare in the West, in regions where HBV remains endemic like Asia, the Western Pacific region and Africa, chronic HBV infection is common and individuals are often first exposed to the virus as infants or children.
Hepatitis B can be spread through bodily fluids such as blood, semen etc. from an infected individual. The most common routes are sexual contact, sharing items (needles, razors, toothbrushes) with an infected person, exposure to infected blood or perinatal transmission (from a mother to child around the time of birth or during breastfeeding). The latter is the main route of transmission in areas where Hepatitis B is endemic like Asia, the Western Pacific and Africa.
The challenge and importance of preventing HBV transmission to infants
The younger an individual is when they contract acute HBV, the higher the likelihood that they will develop the chronic form. This is visualized in the graph below which reflects the percentage of individuals who become chronic HBV carriers in relation to the age at which they were acutely infected. For example approximately 90% of infants who are infected around the time of birth become chronic carriers , in contrast less than 5% of individuals acutely infected as adults are chronic carriers of the disease.
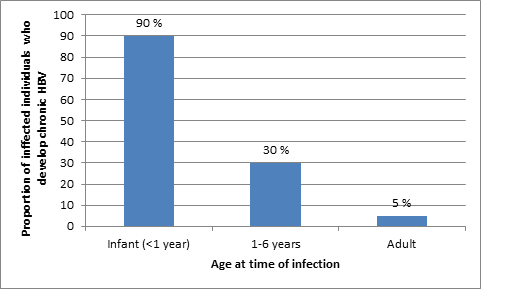
To minimize the risk of infants being acutely infected, babies born to HBV-positive women are regularly given vaccines and other immunoprophylaxis after birth. However 5-10% of vaccinated infants still contract the acute form of the virus. This indicates additional intervention is required to effectively reduce or eliminate transmission to infants.
Infants are an especially critical target population in reducing the prevalence of Hepatitis B, especially since infection rates are still relatively high, and early exposure leads to a higher risk of the disease becoming a chronic life-long disease. For example, long term follow up of individuals infected as infants or young children showed that 15-25% of those chronically infected die prematurely due to cirrhosis or hepatocellular carcinoma.
High HBV activity in the mother correlates with the infant’s risk of contracting the condition, even if the infant has been immunized after birth. Therefore an antiviral therapy which reduces the HBV activity in the mother may be able to prevent the infant from contracting the disease. There are six globally licensed treatments for adults affected by chronic HBV, but none are currently regularly used in pregnancy.
An antiviral drug telbivudine is an attractive candidate for preventing mother to child transmission of HBV. It has been classified as pregnancy category B, which implies that no risk has been shown in other studies, but risk has not been definitively excluded.
Previous studies (1,2) have reported that administration of the antiviral telbivudine in the second and third trimesters was highly safe and effective in blocking mother to infant transmission . However, further extensive studies are required to ensure safety and efficacy before it can be considered for regular use during pregnancy.
The issue of establishing drug safety in pregnancy

It is critical that any treatment given for a maternal medical condition, such as HBV, does not disrupt the baby’s normal process of development or cause significant negative side effects.
This was clearly illustrated by thalidomide, a drug taken by expecting mothers to combat morning sickness. When thalidomide was first introduced as an over the counter drug in 1957, scientists did not believe drugs could cross the placenta and harm the fetus. Unfortunately, this was not the case and it is estimated that over 10,000 children in 46 countries were born with developmental abnormalities (classically of the limbs called phocomelia) as a result of thalidomide exposure.
Although finally taken off the market in 1961, the detrimental effects of thalidomide were long reaching and prompted the implementation of more rigorous rules for the testing and licensing of drugs.
Additionally, tolerability of a drug by the growing fetus may depend on its stage of development- certain drugs may therefore be potentially safe to take in late pregnancy but not during the first trimester when the fetus is very vulnerable to environmental insults.
In terms of HBV treatment during pregnancy, one of the questions which remains unanswered is whether there is a difference in efficacy if antivirals are given to pregnant women during early pregnancy, and additionally whether this can be tolerated by a more vulnerable, less developed fetus. To begin to answer this, Dr. Sun and colleagues recently conducted a study to evaluate the efficacy and safety of telbivudine if commenced in early or middle pregnancy.
Antiviral HBV treatment during early and mid-pregnancy
Dr. Sun and colleagues compared markers of HBV infection and activity in three different experimental groups. One group of women began the antiviral treatment in early pregnancy (12 weeks), another in middle pregnancy (20-28 weeks) while the third group acted as a control and received no drug.
As per standard care, all infants in all groups received standard vaccinations and preventative treatment after birth. At 28 weeks following birth, infants were tested to check for the presence of HBV DNA or HBV antibodies, which indicates maternal to child transmission of the disease had occurred.
The results of the study were clear cut- none of the infants born to mothers who were receiving the antiviral treatment showed biological markers of the disease. In contrast, in the group that did not receive the antiviral during pregnancy, ~18% of infants had markers of HBV activity 28 weeks after their birth.
Similar to previous studies, these results suggest that treating mothers during to pregnancy with the antiviral telbivudine together with infant vaccination at birth appears more effective than infant vaccination alone. Additionally, it suggests antiviral administration may be equally effective if begun in early versus middle pregnancy.
Looking to the future

Although the work by Sun and colleagues showed promising results, it also raises a number of other important questions which would need to be resolved before the antiviral can be used outside of controlled clinical trials. For example, the optimal timing and dosage are still to be determined, as well as how late in pregnancy the antiviral can be given and still successfully prevent transmission. Additionally, while no ill effects were observed in the infants whose mothers received telbivudine during pregnancy, it is important to note that potential negative effects have not been ruled out and more extensive studies with long term follow up are required.
This study provides a critical stepping stone towards the development, implementation and safety of programs which could drastically reduce the perinatal transmission of HBV- and provide infants a healthy start to life.
If you are interested in reading more on this topic, please visit BMC Gastroenterology or the papers below can provide more in-depth information on topics discussed in this blog:
Elimination of Hepatitis B: Is It a Mission Possible?
About Hepatitis B during pregnancy
Maternal hepatitis B virus carrier status and pregnancy outcomes: a prospective cohort study
Effect of elective cesarean section on the risk of mother-to-child transmission of hepatitis B virus
About Immunization
About Telbivudine
Telbivudine versus entecavir in patients with undetectable hepatitis B virus DNA: a randomized trial
Comments