Nematodes – the problem
Nematodes, or roundworms, are a diverse group of organisms that are widely dispersed in nature. While some nematodes, for example the model organism Caenorhabditis elegans, are free-living in the environment, feeding on microbes such as bacteria and fungi, others parasitize plants and animals, and can lead to serious disease in their hosts.
Closely related to hookworm parasites that infect millions of humans worldwide, the strongyles and trichostrongyles are a group of nematodes predominantly found in the gastro-intestinal tract of livestock and includes parasites that cause clinical disease, from weight loss and diarrhoea to anaemia and death. The negative impacts of these parasites can be countered by applying anthelmintic drugs, however the emergence of drug resistance is now reducing their effectiveness as a control measure.
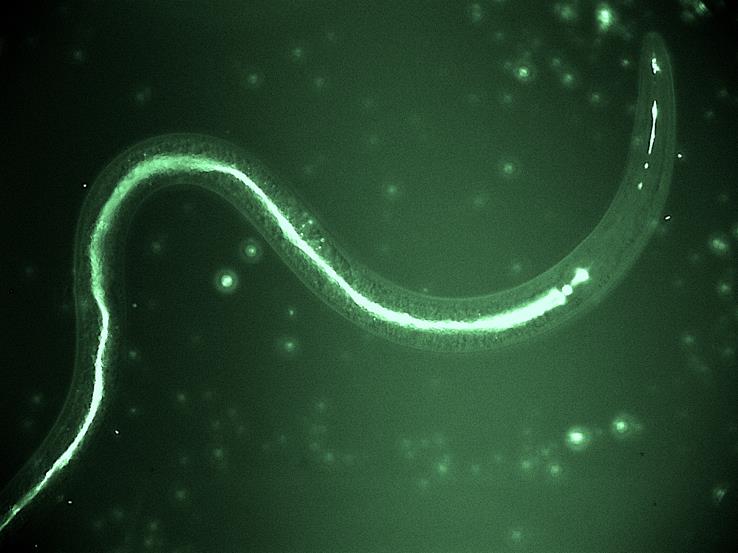
Fighting parasites with parasites
One approach to try and understand nematode biology is to look to the diversity of wild isolates found across the world. For example, C.elegans, can be found in microbe-rich environments associated with rotten plants and fruits. This has proved a rich hunting ground for researchers to identify naturally occurring pathogens, including fungi microsporidia, bacteria, oomycetes and viruses (1, 2).
In our recent study in BMC Biology, we described one such bacterial pathogen, the “golden Death bacillus” Chryseobacterium nematophagum, which we found in a rotten apple in Paris and also from a rotten fig in Bangalore. We characterized how it infects and kills its parasitic worm hosts, and, similar to other natural pathogens, we suggest that C. nematophagum could be a new weapon in the fight against pervasive animal nematode parasites.
Chryseobacterium nematophagum is a formidable pathogen of nematodes
The gram-negative bacterium C. nematophagum we characterized is unusual in many respects; most importantly it is extremely virulent towards a wide range of nematodes that parasitize animals – including the important Strongyle and Trichostrongyle species.
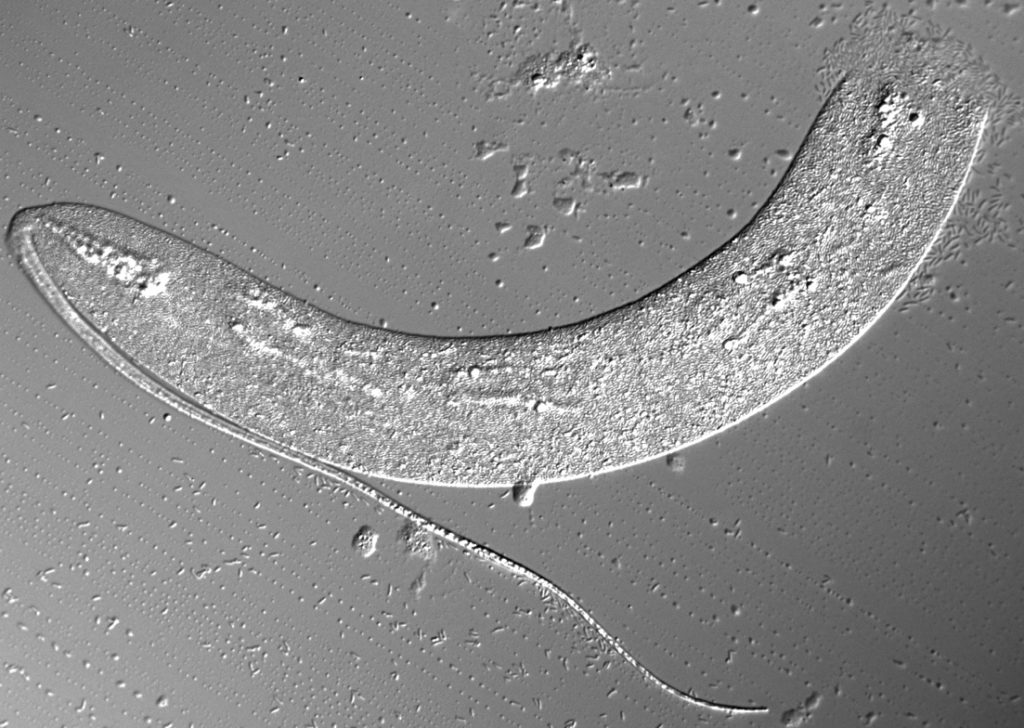
We found that the bacteria kill worms rapidly at a very low dose: within 24 hours when diluted as part of a bacterial food-source, or in just a few hours when given as a pure culture. Unusually, the nematodes are actually attracted to and actively eat these bacteria.
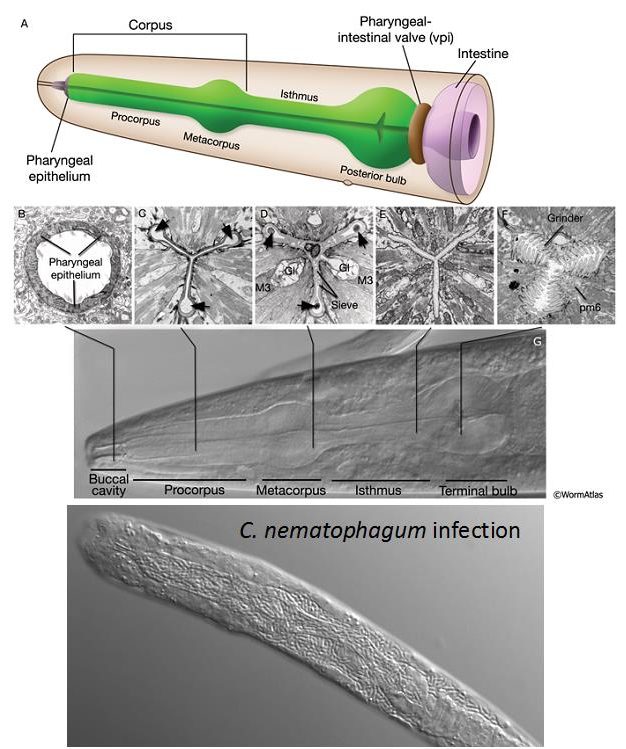
Once ingested the bacteria begin their invasion process, which involves the digestion of the nematode’s anterior pharynx (right), entry and multiplication within the body cavity, and ultimately the digestion of the host from the inside out – leaving just cuticle exoskeleton, but even this resilient structure is ultimately digested. The ubiquity of nematodes in nature is partly down to the fact that they are encased in this normally highly resistant collagenous cuticle.
This fast killing process involves the digestion of the tough pharyngeal structures that include the chitinous and collagenous lining. By comparing the genomes of Chryseobacterium nematophagum to numerous environmental Chryseobacterium species, we identified genes that encode collagenase and chitinase enzymes that are predicted to be involved in this unusual digestion process. These enzymes are found exclusively in Chryseobacterium nematophagum and significantly the substrates for these enzymes do not exist in prokaryotes indicating they are used to infect the nematode host .
The lifecycle of a typical trichostrongyle (such as the important veterinary parasites) involves a free-living phase, where larval stages (L1-L3) feed on environmental microbes prior to developing to the sheathed L3 stage which in turn infects the grazing host. While this newly described nematode pathogen has great potential as a future biocontrol agent, limitations include the fact that it is only effective against the environmental bacteria-feeding stages and therefore control strategies would need to center on the application of this bacteria to the pasture.

However, this approach could be optimized by applying bacteria at seasonal peaks in larvae abundance and perhaps by using the grazing host to distribute the bacteria with their faeces, the location of greatest larva abundance. Finally, the identification and confirmation of virulence factors and invasion associated enzymes may ultimately lead to their potential application in future nematode control strategies.
Tony Page
Latest posts by Tony Page (see all)
- Golden Death Bacillus – the nematode digesting bacteria with biocontrol potential - 28th February 2019
Comments