We asked the authors of Modeling radiation injury-induced dell death and countermeasure drug responses in a human Gut-on-a-Chip to elaborate on the significance of their recent publication in Cell Death and Disease. In the following interview, lead author Sasan Jalili-Firoozinezhad, of the Wyss Institute at Harvard University, provided answers to our questions.
Can you briefly explain what your results show?
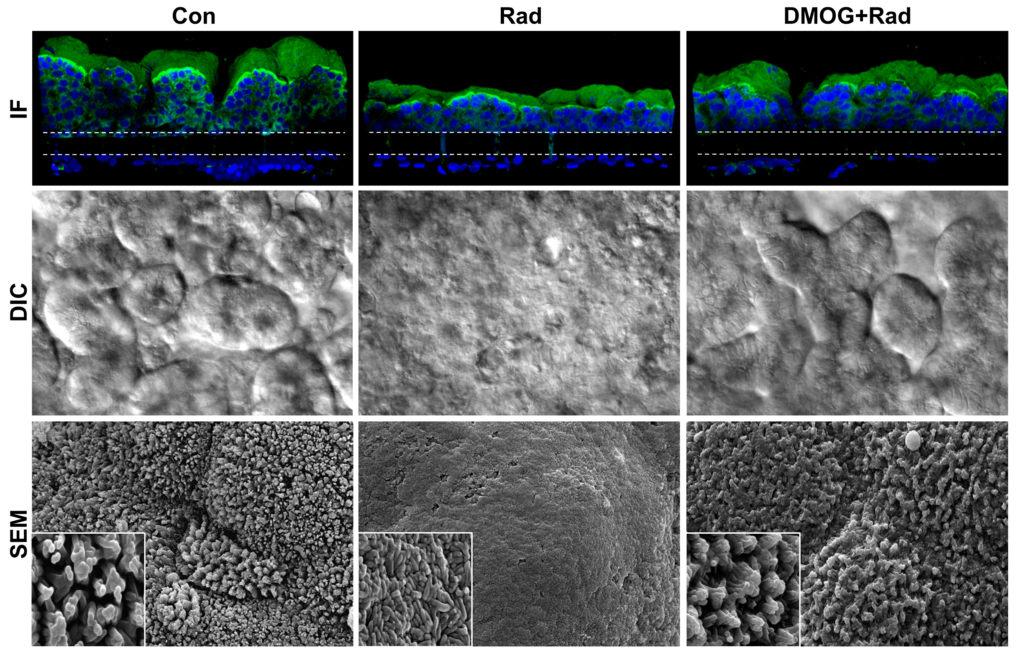
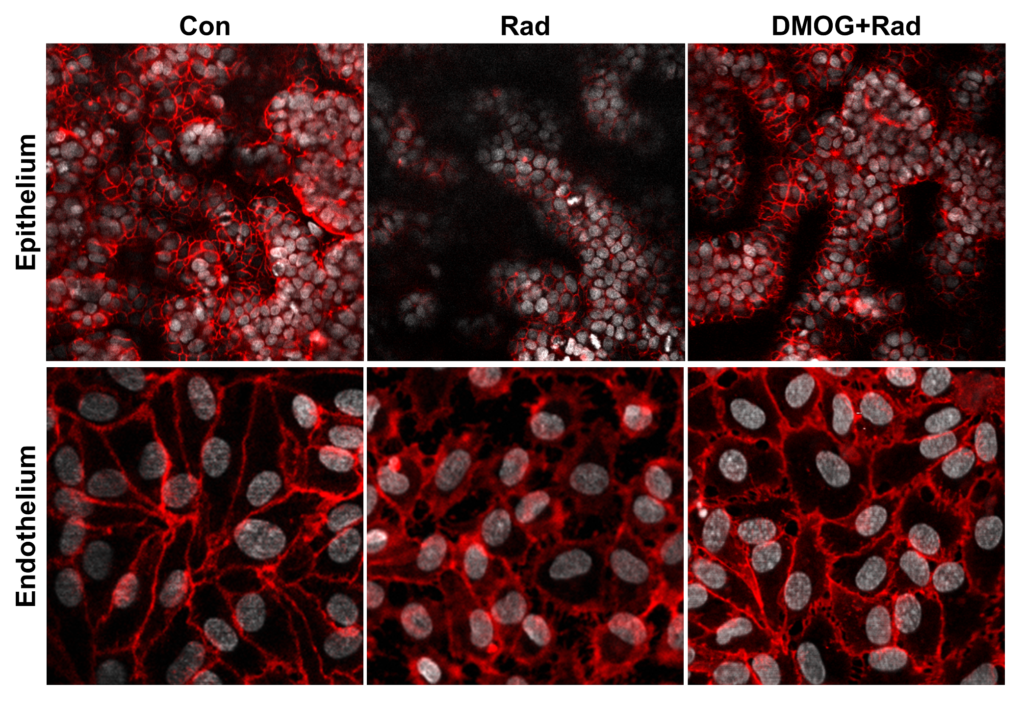
Exposure to ionizing g-radiation, whether therapeutic or accidental, may result in acute radiation syndrome that is associated with gastrointestinal disturbances. Using organ-on-a-chip (Organ Chip) microfluidic culture technology, we developed a model of the gut lined by human intestinal epithelium interfaced with vascular endothelum, which simulated radiation-induced intestinal injury at a radiation dose of 8 Gray (Gy), which is known to cause gastrointestinal effects in humans. We observed increases in several markers of cell damage in both the endothelium and epithelium. These included increased apoptosis (or cell death), generation of reactive oxygen species (ROS, or free radicals), double-stranded DNA breaks, degradation of membrane lipids, and loss of microvilli structure, as well as disruption of cell-cell junctions that form the barrier that protects the intestinal wall from bacteria and toxins.
Using the Gut Chip radiation model, we confirmed that pre-treatment with a potential radioprotective drug, dimethyloxaloylglycine (DMOG), significantly reduced apoptosis, intestinal permeability, and microvillus injury of intestinal epithelial cells, as well as ROS generation and lipid degradation in both endothelium and epithelium, a result that has never been demonstrated in previous experimental models.
Can you briefly explain what the Gut Chip is and why your team elected to make modifications to it for your study?
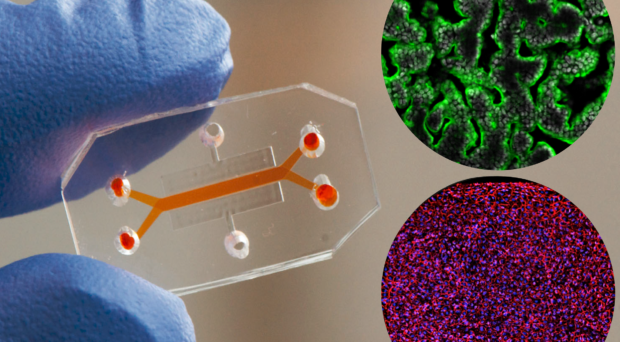
The human Gut Chip is a microfluidic culture device composed of a clear, flexible and transparent silicone polymer about the size of a computer memory stick (see top image). The Gut Chip contains two parallel channels separated by a porous membrane; human intestinal epithelial cells are cultured on top of the membrane in one channel and human microvascular endothelial cells on the opposite side of the same membrane in the second channel. Culture medium is perfused through both channels, and suction is applied to side chambers within the chip at regular intervals to mimic normal cyclic motions in the gut (peristalsis). Within about a week, the Gut Chip simulates the villus structure and multiple functions of the living intestine.
Unlike past radiation models, we incorporated vascular endothelium in the Gut Chip, as recent animal studies suggest that these cells play a key role in the gastrointestinal damage observed in radiation injury. Our results confirm this suspicion: the endothelial cells in the vascular channel exhibited a stronger response to radiation than the epithelial cells in terms of ROS generation, lipid degradation, DNA fragmentation and apoptosis. Interestingly, when we repeated the on-chip experiments without endothelial cells, irradiated intestinal cells did not express their typical response to radiation, suggesting that endothelial cells serve as mediators of radiation-induced intestinal dysfunction.
What are the implications of your study for therapeutics developers, doctors, and patients?
The Gut Chip could be used for discovery and screening of new and more effective countermeasure drugs in the future to save lives of cancer patients, astronauts, or civilians who are accidentally exposed to radiation.
People may be exposed to ionizing g-radiation accidentally (e.g., nuclear accidents, cosmic rays in spaceflight) or as a result of radiation therapy. These accidental exposures can cause cancerous genetic mutations and death, and while radiation therapies use lower doses, they can still cause harmful cumulative side effects. As exposing healthy people to radiation for clinical trials would be unethical, efforts to identify drugs that can mitigate the effects of radiation exposure have been limited to animal studies, which are notoriously poor predictors of how a given drug will behave in humans. The human Gut Chip radiation model, however, offers a potentially powerful tool for studying the mechanisms that underlie radiation-induced injury. The Gut Chip could be used for discovery and screening of new and more effective countermeasure drugs in the future to save lives of cancer patients, astronauts, or civilians who are accidentally exposed to radiation.
What future directions do you see for your research based on your results (maybe elaborating on the ‘human Body-on-a-Chip’ idea)?
When animals or humans are exposed to total body radiation doses that lead to severe injury, lethality is mainly due to the combined effect of damage to the gastrointestinal tract, lung, and bone marrow. For this reason, we are also studying radiation injury using human Organ Chip models of these organs. In addition, there is evidence to suggest that radiation injury of the gut, for example, can influence the degree of damage experienced by the bone marrow in the same individual in a manner that is influenced by the presence of the gut microbiome. Thus, we have developed methods to culture complex communities of commensal microbes that comprise the human gut microbiome in our Gut Chip, which will allow us to analyze how the microbiome contributes to these responses.
…we are linking different human Organ Chips via their endothelium-lined vascular channels to effectively create a human ‘body-on-chips’…
In addition, we are linking different human Organ Chips via their endothelium-lined vascular channels to effectively create a human ‘body-on-chips’ that could potentially be used to model these organ-organ interactions that mediate human responses to radiation exposure. To enable development of personalized body-on-chips models, we also have created Intestine Chips with cells isolated from patient biopsies, and others have shown this can be done using patient-derived induced pluripotent stem (iPS) cells as well. We are now leveraging these novel models in combination with bioinformatics and computational discovery approaches to identify new radiation countermeasure drugs.
Comments