
It is thought that gene duplication plays an important role in generating new genetic material for the evolutionary diversification of species. Gene duplication can be caused by several mechanisms and in the extreme case two copies of all of the genes in a genome can be generated by whole genome duplication (WGD).
In a recent study published in BMC Biology, my colleagues and I analyzed the genome of the common house spider Parasteatoda tepidariorum and we have found evidence for a WGD in the lineage leading to spiders.
This event was likely shared with scorpions and probably other arachnopulmonates like whip-scorpions and whip spiders, but not with more distantly related arachnids such as ticks and mites. This suggests that the approximately 45,000 extant species of arachnopulmonates evolved from a polyploid ancestor over 400 million years ago.
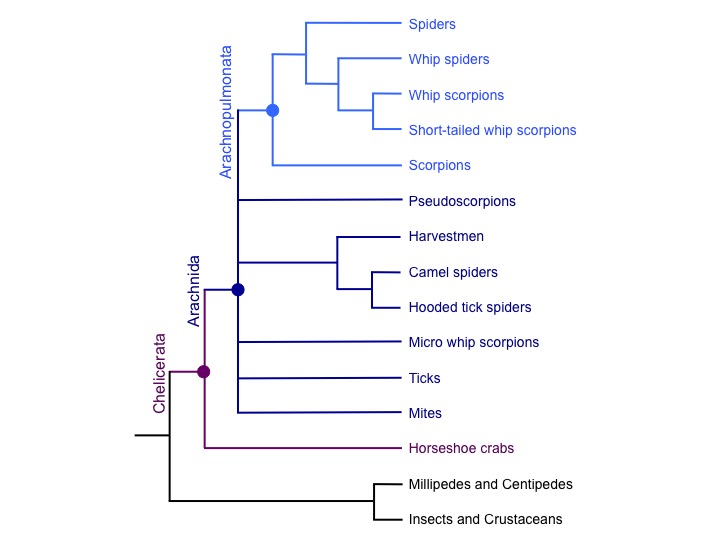
Our study also suggests that this WGD in arachnids was likely independent of WGD in another group of chelicerates, the horseshoe crabs. Our findings thus offer an exciting opportunity to discover more about the outcomes of WGD in terms of gene content and regulation, and how such events may contribute to animal diversification.
In the ancestor of vertebrates there were two rounds of WGD and these events may have led to the diversification of these animals through the retention and utilization of duplicated genes. For example, while most animals contain a single cluster of Hox genes, vertebrates, like ourselves, have four Hox clusters and these additional genes play many important roles in development. However, genes can be retained or lost for many reasons after WGD events, including random mutation, recombination and dosage effects.
Identifying whole genome duplication events
A better understanding of the patterns of gene retention and loss after WGD and identification of commonalities, as well as potentially the genes that may underlie evolutionary innovation, requires the study of independent events. This comparison, however, suffers from the fact that only a few examples of WGD have been described in animals to date.
Our identification of a WGD in arachnids, consequently, provides a much-needed new data point for understanding the general and lineage specific impact of WGD events.
Our identification of a WGD in arachnids, consequently, provides a much-needed new data point for understanding the general and lineage specific impact of WGD events. The duplicated genes that have been retained in both spiders and scorpions represent many that encode proteins with important roles in development, including two copies of most Hox genes arranged in two (nearly) complete clusters.
Furthermore the paralogs of each of the spider Hox genes differ in their timing and spatial expression during embryogenesis suggesting that some of the new copies perform novel functions with respect to the single copy ancestral gene. Therefore our study reveals an intriguing parallel between the outcomes of WGD in arachnids and vertebrates.
Interestingly, our study and previous work also reveals a high rate of retention of duplicated microRNAs. These genes are thought to modulate the expression levels of their target genes and they have perhaps been retained in high numbers after WGD to buffer the dosage effects of targeted duplicated protein coding genes, rather than contributing to the emergence of novel traits. Indeed, a possible outcome of gene duplication is developmental systems drift, whereby different genes and interactions can be used to achieve the same phenotypic outcome, but this remains to be investigated systematically.

More fully understanding the consequences WGD event in arachnids requires the analysis of additional arachnid genomes to determine exactly when this event occurred and which lineages were affected. For example, it would be interesting to explore whether there is any evidence for WGD in other arachnid orders like camel spiders, harvestmen and pseudoscorpions.
In addition, comparing the genomes of whip spiders and whip scorpions with spiders and scorpions could help reveal genes that have been retained by most groups after WGD versus lineage specific retentions and losses. These data will not only provide insights into arachnid genomes ‘before’ and ‘after’ the WGD event, but a better understanding of how duplicated genes produced by this event have contributed to the evolution of innovations in these animals, for example, silk production in spiders and the booklungs (novel breathing organs) of arachnopulmonates.
Finally, a more detailed understanding of the patterns of gene retention and loss after WGD in arachnids will provide an excellent comparison to such events in vertebrates to better understand the broader implications and consequences of WGD for the evolution of animal genomes and their biology.
Comments