The use of animal models has been a vital part of research in many scientific studies. Animal models are able to give ‘in vivo’ data, which is truer to life than that which laboratory conditions (in vitro) would give as they provide researchers with a better understanding of the complexity of biological processes taking place in situ. However, the use of animal models in research brings up important ethical issues and usually increases costs and study time.
The technique of two-dimensional cell culture has provided an easy and efficient way of studying human tissue cultures, parasites and pathogens in the laboratory. However, a problem with this kind of culturing is that the cells are in contact with an artificial surface and adopt a planar shape on the plate, which can affect important aspects of the biology of the cells. In contrast, three-dimensional cell culture could provide a microenvironment that is more natural and enable cell architecture to resemble the in vivo situation.
The use of hydrogels for 3-D culturing
Hydrogels are networks of natural or synthetic polymer chains that are hydrophilic. They can absorb a large amount of water which makes them very flexible, hence their use in products such as nappies. Hydrogels are able to hold cells in a three-dimensional structure without affecting their morphology, and can be made up when needed. They are quick setting, easy to dispose of and relatively cheap, potentially making hydrogels a far better option for culturing cells than using animal models.
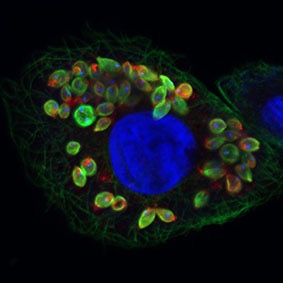
Researchers at Keele University, UK are beginning to investigate the use of hydrogels for the in vitro culture of Leishmania, a unicellular parasite that infects humans.
Leishmania parasite
Leishmaniasis is a vector-borne tropical disease caused by protozoa of the Leishmania genus. It is spread through the female sandfly (phlebotominea) vector.
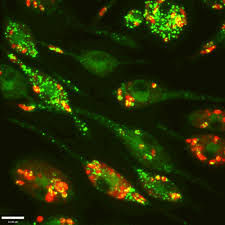
My summer project
During the summer of 2017 I began investigating the formation of hydrogels, and their use for culturing THP-1 macrophages in the Centre for Applied Entomology and Parasitology at Keele University. I found that collagen constructs, used as the main constituent for the formation of hydrogels were suitable for the culturing of these cell lines. However, I found that the macrophage cell lines needed to be pre-mixed in the gel during its formation. Investigation of the literature revealed that PMA (phorbol 12-myristate 13-acetate), which is used to stimulate differentiation of the cells, affected the collagen structure of formed gels when the cell lines and PMA were placed on top of the already formed gel for an ‘invasion’ analysis.
I also discovered that high-resolution imaging equipment must be used for imaging of the gels, as an epifluorescence microscope was unable to give clear images of the cells within the gel. Blurry and dull images were caused by light being scattered by the collagen fibers before reaching the eyepiece of the microscope. Techniques such as confocal, or multiphoton microscopy are therefore required for future work.
Researchers hope to be able to show that hydrogels can be used for studies that involve the parasite Leishmania by culturing macrophages in this 3-D system, then infecting the cells with the parasite. It is unknown whether there will be a difference in anti-parasitic drug effects, or cell infection rates when 2D or 3D cultures are used. However, work is now being carried out to find the effects of culturing cells and parasites in hydrogels, and comparing the results to 2-D plate cultures.
If there is an improvement in growth rates, drug interactions or cell-parasite interactions, it may be possible for hydrogels to be used as an alternative to both conventional culture systems and animal model studies. Hydrogels are now routinely used to culture other eukaryotic cell types and we may be able to adopt procedures from other research areas. Multiple types of hydrogels can be made, including polystyrene and gelatine as these scaffolds are also able to hold cells in three dimensions. This emerging technique will undoubtedly return some interesting results for the culture of Leishmania-infected macrophages, and might be able to replicate ‘in vivo’ conditions without the use of an animal, giving researchers the ability to mimic parasitic infection and study more effective treatments.

Comments