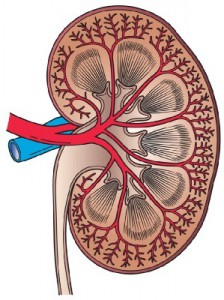
 Glandular organs, such as the liver, pancreas and kidney, contain systems of branched epithelial tubes that resemble trees. The development of these tree structures has been the subject of much previous research and is known to be regulated by factors including signalling molecules and extracellular matrix molecules.
Glandular organs, such as the liver, pancreas and kidney, contain systems of branched epithelial tubes that resemble trees. The development of these tree structures has been the subject of much previous research and is known to be regulated by factors including signalling molecules and extracellular matrix molecules.
In the kidney the urinary collecting duct tree is involved in the movement of urine through the kidneys to the ureter. Researchers from the University of Edinburgh, in new research published in BMC Developmental Biology, explored the development of the urinary collecting duct tree to further analyse how these branches form.
Using cultured kidney rudiments the researchers initially discovered that the position of new branches appeared linked to the distance from other branches. The new shoots grew at angles to ensure the maximum distance between branches with the branches avoiding each other as much as possible. This implied that a ‘repellent’ signalling molecule might be involved in this process and that a mechanism of self-avoidance was driving this process .
A model of a simplified kidney was then used to assess whether this hypothesis was feasible. The model assumed that tree tubules, the beginning of a new branch, secrete a theoretical factor (which the researchers named horrid) which diffuses away from the tubule, decreasing in concentration. Tree branching was assumed to occur only when the concentration of horrid was below a certain threshold and that the branch would then grow in the direction of the lowest horrid concentration. The model produced realistic looking trees, mimicking the patterns seen in nature, and as a result potential horrids were investigated.
The TGFβ-superfamily was considered an obvious place to start as members of this family of regulatory proteins have previously been shown to play a role in other branching systems. In addition treatment of urinary collecting duct cell lines with TGFβ has been shown to inhibit growth. However TGFβ itself is not expressed during the key stages of urinary collecting duct tree branching and as a result was unlikely to be involved.
There are several members of the TGFβ-superfamily expressed at the correct time, including various members of the bone morphogenetic proteins (BMPs) group of growth factors. Of these BMP7 seemed the best candidate due to the fact it is strongly expressed at the branch tips throughout kidney development.
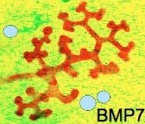 Beads were soaked in BMP7 or a control substance and placed near growing kidney rudiment buds. Buds next to the control beads showed normal growth patterns whilst the buds near the BMP7 beads were truncated or the branches moved away from the beads. This result was confirmed with a chemotaxis assay confirming that BMP7 plays a role in determining the pattern of urinary collecting duct tree branching.
Beads were soaked in BMP7 or a control substance and placed near growing kidney rudiment buds. Buds next to the control beads showed normal growth patterns whilst the buds near the BMP7 beads were truncated or the branches moved away from the beads. This result was confirmed with a chemotaxis assay confirming that BMP7 plays a role in determining the pattern of urinary collecting duct tree branching.
However we know from previous work that mice lacking BMP7, whilst displaying kidney abnormalities, do show some branching indicating that there are other factors in this process. Quite what these factors are and how they interact with BMP7 will be the subject of future work.
Comments