
Where are we now?
As of July 2020, over 19,600 studies have been registered on ISRCTN from organizations in 128 locations. For studies that completed more than a year ago, approximately 58% have results posted to the record. Over 5900 updates were made in 2019.

For users
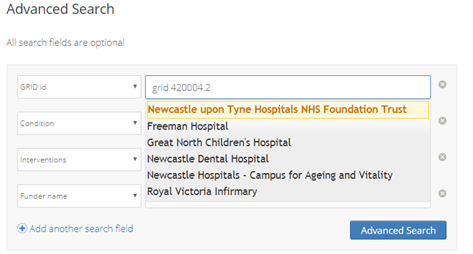
Users are now able to download search results as a CSV file and can select the fields they want to export. The sponsor names have been matched to Global Research Identifier Database (GRID) IDs and suggested matches are displayed at submission so that trialists can select the main name.
This enables more consistent searching – previously the sponsor names were not validated so there was variation in complex names, plus if an organisation changed its name, the two names were not linked. Users are able to search by GRID ID for sponsors and Funder Registry (formerly FundRef) ID for funders.
The copyright licensing has been clarified to emphasise that most of the information in a study record is considered metadata and can be reused without restriction, with defined fields reusable under a CC-BY licence.
Several updates have been made in order to comply with the WHO’s International Standards for Clinical Trial Registries, which came into force at the end of January 2019. These included requiring applicants to confirm that they understand their responsibilities as the registrant, adding a field for the final enrolment number, and restricting the ability to hide study and sponsor contacts’ telephone numbers and email addresses to editors.
For trialists
The WHO expects that all study records should be updated at least once a year until results are published and that basic results or a results article link must be posted to the registry record within 1 year of the completion date.
Researchers can now use buttons labelled ‘Update your record’ and ‘Report your results’ to request these updates. They do not need to log in or remember the registry email. Someone other than the original applicant or the study contacts can use the buttons, which allows sponsor representatives or trial administrators to request an update.
The response to ISRCTN reminder emails continues to be positive. In 2019, nearly 6000 prompts to update records were sent and approximately 1500 updates made in response. This included 438 updates to records with completion dates in 2012-2016 but no results posted.
ISRCTN editors can now directly edit the text in webpages such as the FAQs and field definitions, which means that policy changes can be rapidly communicated. Banners on the ISRCTN home page and submission page enable us to provide relevant updates.
We have recently updated the pop-up guidance in the submission form, giving real-time help when trialists are entering the study information, which seems to have improved the completeness of the initial submissions.
We have also added information in Arabic, Chinese, French and Spanish, including why a study should be registered and what to expect when registering a study on ISRCTN.
The @ISRCTN Twitter account sends daily tweets to draw attention to recently registered studies and ISRCTN-registered studies with recently published results. Follower numbers, referrals and social media engagement are steadily growing.
For the public
In 2019, we replaced the National Institute for Health Research (NIHR) condition indexing with more descriptive terms that would be more likely to be searched by public users for all actively recruiting NIHR studies and those yet to start recruiting.
On 20th May each year, we celebrate Clinical Trials Day by creating fun and informative content on clinical research. This year we focused on COVID-19 – our COVID-19 research quiz was very popular!
Next steps
Development is underway as of July 2020 that is expected to deliver updates to the registry website by September. One of the main items is to enable searching for study records by sponsor country and for UK studies, to enable searching by UK nations in the sponsor country and recruitment country fields. The GRID and FundRef IDs will be made visible.
Another goal is to make the trial setting information more useful to potential participants and other users. We have received feedback that potential participants would like more information on the travel burden involved and to search for studies that can be undertaken without significant travel involved.
Do you have feedback that will help us improve the ISRCTN registry? We would love to hear from you!
Currently, trialists can only select one setting from a short list. However, this does not reflect the reality of many clinical studies where a participant might be recruited by their GP or other primary care provider, visit a hospital to undergo a procedure, then undergo the intervention at home or in the community (for example, exercise as rehabilitation from orthopaedic surgery), and subsequently be followed up at a university.
We intend to expand the list to include settings such as care home, dental clinic, workplace and fitness/sports facility, and to allow multiple settings to be selected, which would give the potential participant a better understanding of the logistics involved.
Do you have feedback that will help us improve the ISRCTN registry? We would love to hear from you! Please contact claire.veryard@biomedcentral.com.
Comments