
Susceptibility to infection in Hymenoptera
Several regions of the world have reported a decline in wild bee species. A contributing factor may be pathogen spillover from managed honeybees, and a variety of pathogens so far exclusively linked to honeybees have been detected in several wild bee species.

However, the detection of a pathogen does not necessarily mean the infection is spreading per se. Pathogens may behave differently within a novel host, where a species may not serve as a suitable host or the parasite does not replicate. Disease progression and symptom intensity can also differ both between and within species.
Several factors influence host susceptibility to pathogens and infection, such as life history traits (e.g. reproductive strategies and sociality), environmental traits, nutrition and the genetics of the host and pathogen. For example, greater infection resistance is typically observed in hosts with genetic heterozygosity and in social groups with higher genetic diversity. The haploid susceptibility hypothesis predicts in Hymenoptera the haploid males from unfertilized eggs should have higher susceptibility (compared to diploid females) as they lack heterozygosity.
Are bee parasites picky parasites?
Trypanosomatids are unicellular eukaryotic flagellate parasites, and some species colonize the hindgut of bees. They damage intestinal cells through formation of hemidesmosomes, and these lesions can reduce host health at the individual and colony level. In bumblebees, parasite cells multiply after oral ingestion and are transmitted to novel bumblebee hosts via fecal-oral transmission.
The trypanosomatid parasite Crithidia mellificae infects honeybees. While little is known about the effects of infection, there are positive correlations between C. mellificae infection levels and honeybee colony winter mortality. Two common solitary bee species, Osmia cornuta and Osmia bicornis have also recently been suggested as novel hosts of C. mellificae.
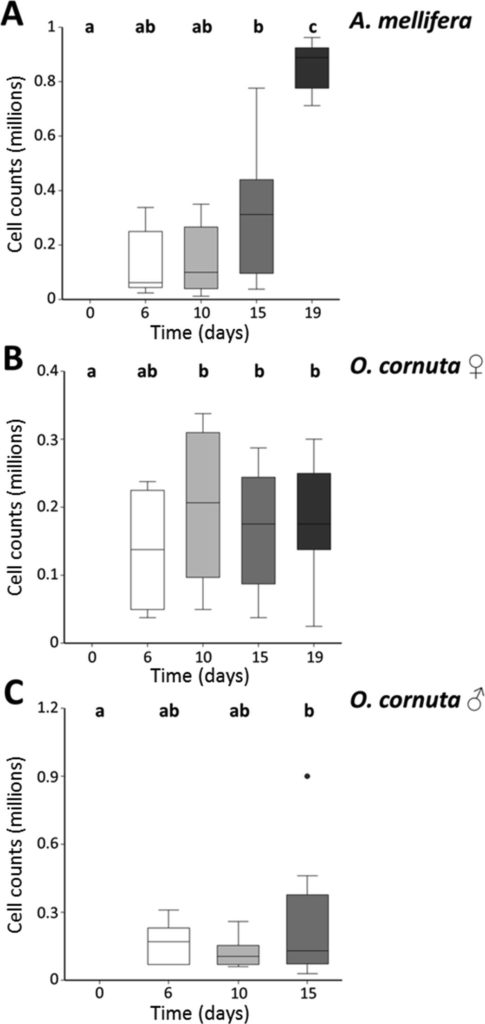
In a recent paper, Strobl and colleagues tested whether this parasite could infect a solitary bee by challenging male and female O. cornuta with C. mellificae. The authors also employed honeybee workers as positive controls to assess infection parameters in a known host. Bee body mass, survival and pathogen infection levels were evaluated as measures of susceptibility.

The experiment
O. cornuta were randomly allocated to treatment (exposed to C. mellificae cells) or uninfected controls, and the males and females separated (control: n = 2 cages, C. mellificae exposure: n = 4 cages). Honeybee workers were randomly allocated to eight cages (control: n = 3, C. mellificae exposure: n = 5), and all cages were maintained for 19 days.
Each cage was provided with a C. mellificae-infected sucrose solution or with sucrose (controls). Survival was recorded every 24 hours and dead individuals were removed. Bees were investigated for living C. mellificae cells before inoculation with C. mellificae (day 0) and on days 6, 10, 15 and 19 post inoculation (p.i.). Bees were individually weighed to assess body mass and anesthetized with CO2, before quantifying C. mellificae cells. DNA sampled on day 0, 15 and 19 was analyzed by PCR and parasite loads were quantified by qPCR.
Results
In all groups of bees, body mass did not significantly change over time p.i. (all P> 0.05) or differ between control and C. mellificae–exposed individuals.
After 19 days p.i., 75.5% of the 83 control honeybees and 63.2% of the 128 C. mellificae-exposed honeybees were alive, with significantly reduced survival in parasite-exposed individuals. Of the 80 honeybees sampled for C. mellificae counting, 32.5% of the bees were infected. Parasite cell counts increased 6.6 fold in infected honeybees between days 6 and 19 p.i. and significantly reduced survival.
C. mellificae exposure did not significantly affect survival of O. cornuta females. Of the 25 control O. cornuta females 80.7% survived compared to 68.1% of the 56 C. mellificae-exposed females. However, male O. cornuta had the lowest survival of all bee groups. After 19 days p.i., 39% of the 43 controls and none of the 81 C. mellificae-exposed male bees were alive. Of the 41 O. cornuta females sampled over the entire p.i period, 68.3% showed C. mellificae cells, whereas 90% of the 30 males sampled showed parasite cell counts.
In O. cornuta bees, C. mellificae numbers increased 2–3.6 fold within cages but did not significantly change between days 6 and 19 p.i. in individual females or males. In both female and male O. cornuta, the proportion of infected individuals increased over time p.i.
A significant positive correlation between C. mellificae cell counts and C. mellificae genomic equivalent copies per bee was also observed in all groups of bees.

A walk on the wild side for trypanosomatids
Strobl and colleagues demonstrate that the solitary wild bee O. cornuta can host the honeybee parasite C. mellificae, and that males are more susceptible. The proportion of infected hosts also increased in O. cornuta cages with feces, but not in honeybee cages without feces. This indicates a fecal-oral transmission route for C. mellificae, as with Crithidia species infecting bumblebees.
The reduced survival of infected honeybee workers provides some causal evidence for the correlation between overwintering colony mortality and C. mellificae infection levels. With evidence of a negative effect on managed honeybee species and male wild solitary bee species, field studies are required to evaluate spillover potential from managed to wild bees and vice versa.

Comments