Written by Professor Bryan Turner, University of Birmingham, UK
The May 1964 issue of the Proceedings of the National Academy of Sciences USA, contained a paper entitled “Acetylation and methylation of histones and their possible role in the regulation of RNA synthesis” by Allfrey, Faulkner and Mirsky, working at the Rockefeller Institute (now Rockefeller University) in New York (Allfrey et al. 1964). The paper presented evidence that RNA synthesis in vitro was inhibited less effectively by chemically acetylated histones than by native histones. Based on this, the authors suggested that acetylation might be a means of “…switching-on or –off RNA synthesis” . They were appropriately cautious in proposing this conclusion (“…the evidence supporting this view is, at best, tentative…”), but we now know that it is essentially correct; histone acetylation, acting in concert with other modifications, is indeed an important means of regulating gene expression. The paper is rightly seen as a landmark in our appreciation of the crucial role of histone modifications and is frequently cited. However, whether histone acetylation itself can ever be the sole switch that turns genes on and off remains contentious, as do the mechanisms by which it operates. If we are to properly celebrate this paper and its place in the history of chromatin and epigenetics, it’s worth taking a closer look at the experiments it contains.
Fifty years ago, it was by no means clear what the histones actually did in the nucleus. It was known that they were present in large amounts, comparable by weight with DNA, and that they were highly positively charged, and therefore bound tightly to the negatively charged DNA. It was suggested that they might be involved in DNA packaging or suppression of RNA synthesis on the DNA template, but the experimental evidence supporting these ideas was very limited. Knowledge about the composition of the histone protein family and the properties of the different members was also limited. Histones could be fractionated by biochemical means into arginine-rich and lysine-rich fractions, allowing amino acid content to be analysed and the amino terminal residues to be identified. It was known that, unlike most other proteins, the alpha-amino group of each histone’s N-terminal amino acid was usually acetylated (“blocked”), but there was little, if any, evidence for acetylation of internal lysines.
In the 1964 paper, Allfrey and colleagues set out to explore the modification of histones by acetylation and methylation, to confirm that histones could inhibit RNA synthesis in vitro and to ask if histone acetylation could alleviate this inhibition. The paper showed that radiolabelled acetate (from sodium acetate-2-14C) was incorporated into histones by isolated calf thymus nuclei, even when protein synthesis (there must have been some residual cytoplasm attached) was inhibited by puromycin. These experiments show that incorporation of acetate occurs after synthesis of the protein chain, an important finding. (In later papers, the group identified the radiolabelled amino acid as Ɛ-acetyl lysine). The paper then describes how added histones inhibited the synthesis of RNA (14C-ATP incorporation) in both a crude extract of calf thymus nuclei, using endogenous polymerase and DNA, and partially purified preparations of bacterial polymerases. Finally, histones were chemically acetylated with varying concentrations (over a 100-fold range) of acetic anhydride, and it was shown that the acetylated histones were less efficient inhibitors of 14C-ATP incorporation under the test conditions than their untreated counterparts. The authors emphasized that the acetylated histones were still highly positively charged and still able to bind to DNA and increase its melting temperature, suggesting that the acetylation, even at high acetic anhydride levels, had affected only a minor proportion of available amino groups. The implication was that the effect of acetylation was not due simply to a gross change in net charge and a general relaxation of DNA binding.
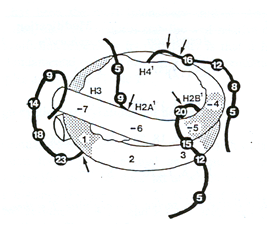
There is little to be gained from critical analysis of these experiments 50 years on; the technical limitations were clear even at the time they were done, and the authors were suitably cautious in the wording of their interpretations. But they remain remarkable for how far ahead of their time they were. They predated the growing understanding of the role of histones in DNA packaging and gene regulation by at least ten years. The famous beads-on-a-string electron micrographs of spread chromatin (Don and Eva Olins) appeared only in 1974, while the low-resolution structure of the nucleosome from X-ray crystallography (Klug et al) came only in 1980 (Figure 1). This allowed a much clearer understanding of the location of potentially acetylated lysines and how they might interact with DNA. More recently has come the realisation that acetylation of individual histone lysines can exert specific functional effects, sometimes through selective binding of proteins through their bromodomains. The current model sees the nucleosome, and its many modifications, as a subtle and complex signalling module by which the genome responds to external cues (reviewed in (Turner 2012)). In this respect it is worth quoting Allfrey and Mirsky’s interpretation of their data in their abstract to the National Academy of Sciences annual meeting in Washington D.C., April 1964. “These findings raise the possibility that relatively minor modifications of histone structure, taking place in the intact proteins, offer a means of switching RNA synthesis on or off at different loci” (my underlining).
The Allfrey et al. experiments tell us little about the mechanisms by which histone acetylation regulates gene expression, which even now are only gradually being revealed. However, the work demonstrated the post-translational acetylation of histones and drew attention to its possible role in regulating RNA synthesis at a very early stage in the field’s development. In subsequent work the Rockefeller group, and Vince Allfrey in particular, maintained this focus as the chromatin field gradually came to appreciate the importance of the topic they had drawn attention to in 1964. This alone makes the 50th anniversary worth celebrating.
REFERENCES
Allfrey VG, Faulkner R, Mirsky AE. 1964. Acetylation and Methylation of Histones and Their Possible Role in the Regulation of Rna Synthesis. Proc Natl Acad Sci U S A 51: 786-794.
Turner BM. 2012. The adjustable nucleosome: an epigenetic signaling module. Trends Genet 28: 436-444.
Sam Rose
Latest posts by Sam Rose (see all)
- Raising funds for genetic diseases - 23rd September 2016
- The Epigenetics and Chromatin Clinic - 9th November 2015
- Resurrecting one of the oldest genetics journals - 23rd October 2015
Comments