
Despite working around the clock to respond to an unprecedented public health crisis, the hard-working staff at the FDA’s Center for Drug Evaluation and Research (CDER) is committed to rare disease drug development for patients. Throughout the challenges we all faced in 2020, CDER continued to build on our previously successful years and approved 31 new molecular entities (NMEs), also called novel drugs (those never before approved or marketed in the U.S.) with Orphan Designation. That’s 58% of all new drug approvals in CDER for 2020! In fact, as the chart below shows, from 2010 through 2020, 190, or 44%, of our 431 novel drug approvals were approved to treat patients with rare diseases.
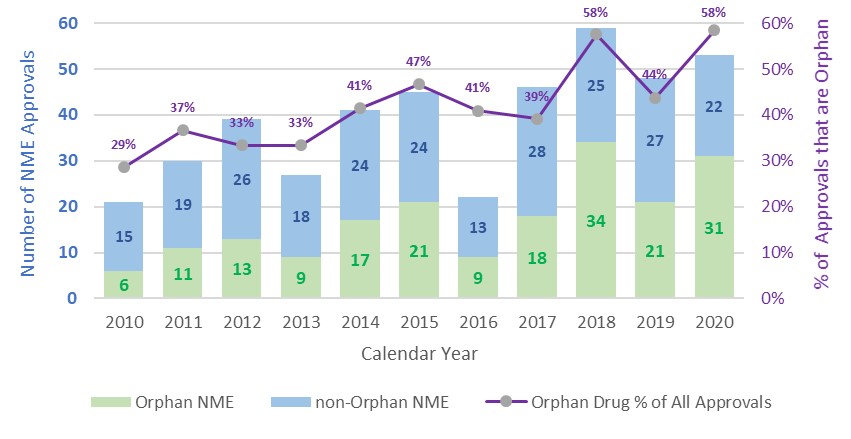
Our novel approvals tell a big part of the story, but every year CDER also approves many additional uses for already-FDA approved drugs to help patients with rare diseases. These are called supplemental approvals. Our novel and supplemental orphan approvals will help a wide range of patients suffering with serious and in some cases, life-threatening conditions to have better quality of life, improvement of symptoms, and in some cases, longer lives. This year’s orphan approvals spanned advances in oncology, neurology, infectious diseases; and other rare disorders across our now 27 review divisions in the Office of New Drugs. Some highlights of our 2020 approvals were:
- Therapies to help patients with Hutchinson-Gilford Progeria Syndrome and progeroid laminopathies, thyroid eye disease, hypereosinophilic syndrome, a new oral therapy for spinal muscular atrophy, two new drugs to treat Ebola virus, and two new treatments for patients with deficiency of interleukin-1 receptor antagonist.
- In cancer disorders, CDER approved three treatments for patients with multiple myeloma, one to treat high-risk refractory or relapsed neuroblastoma, two to treat gastrointestinal stromal tumors, and a new drug for neurofibromatosis type 1, among many others.
- For a full list of CDER novel approvals and many examples of important supplemental approvals for rare diseases, please see our annual report, Advancing Health Through Innovation: New Drug Therapy Approvals 2020.
How do we do it?
Collaboration, communication, a strong foundation in science and drug regulation, and our unwavering commitment to patients are our primary tools. First and foremost, our dedicated multidisciplinary review staff including clinical, biostatistics, clinical pharmacology, pharmacology/toxicology, and regulatory experts review and evaluate the data, often on priority (shorter) review clocks. Note that many of our Orphan NMEs meet requirements for and are granted use of our expedited program pathways which allow us to prioritize resources and timelines to aid in their development and review.
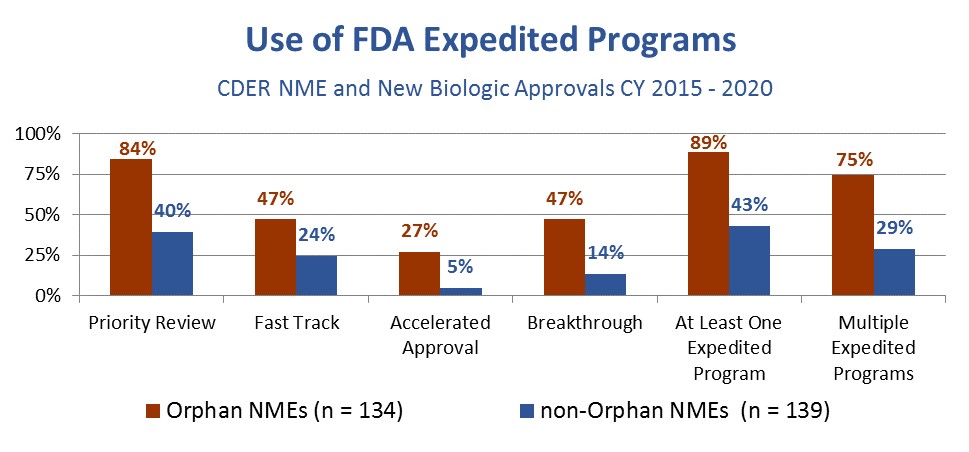
Our expedited programs accelerate development and shorten timelines for review of qualifying applications — often resulting in important drugs reaching patients sooner than if reviewed via standard procedures. In 2020, 29 of our 31 novel orphan approvals, 94%, used one or more expedited program.
Looking Ahead
CDER is very excited about our ongoing efforts to further enhance drug development for rare diseases. Drug development can be challenging for drug products in this space and we have created new tools.
Patients are our most important partners in rare disease drug development
Within the Office of New Drugs (OND), a re-organization in 2020 created a new OND rare diseases hub in the Division of Rare Diseases and Medical Genetics, which is comprised of two collaborative groups focusing 100% on rare diseases: inborn error and medical genetics expert review staff assessing drug development programs and applications, and a multidisciplinary Rare Diseases Team (RDT) to coordinate OND’s rare disease policy and programmatic functions such as developing guidances, educational training, and work with multiple stakeholders, both internal and external to FDA. The Division’s mission is to facilitate, support, and accelerate the development of drug and biologic products for the benefit of patients with rare diseases.
To support innovation and quality in the drug development pipeline for rare diseases, FDA established a “Rare Disease Cures Accelerator”. Through this program FDA is working to facilitate a cooperative approach and common standardized platforms to better characterize rare diseases, incorporate the patient’s perspective in clinical outcome assessment measures (which are used in clinical trials to test if a drug works), and build clinical trial readiness in the pre-competitive space. Patients are our most important partners in rare disease drug development, and this project looks to combine their collective power into a data platform accessible by all to inform drug development as well as develop new patient-focused measures applicable for use across multiple therapeutic areas.
We hope this overview provides you with an understanding of CDER’s ongoing commitment and excitement about our continued support for patients with rare diseases and our efforts to advance development of safe and effective therapies to treat their conditions. To recognize Rare Disease Day 2021 and engage with the rare disease community, we invite you to attend FDA’s virtual public meeting on March 5, 2021, to bring together stakeholders to highlight strategies to support rare disease product development.
Comments