CONSORT and reporting trials in 2017: who should do what
In celebration of Clinical Trials Day, we invited the CONSORT Group to reflect on what they see the future holding for clinical trial reporting. Here, Larissa… Read more »

In celebration of Clinical Trials Day, we invited the CONSORT Group to reflect on what they see the future holding for clinical trial reporting. Here, Larissa… Read more »

The CONSORT guidelines outline the minimum reporting items that should be included for the complete and transparent reporting of randomized controlled trials.… Read more »

Is it time for a transparency metric? Scholarly metrics are thriving right now, both in number and what (and how) they measure, there is a real interest in them… Read more »

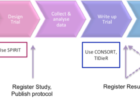
This post was co-written with David Moher, co-Editor-in-Chief of Systematic Reviews. While much of the focus on reducing waste in research has been on primary… Read more »