BioMed Central recently convened some inaugural ‘threaded publications’ discussions in Oxford, UK. This post is an expanded summary of the discussions and next steps for the concept. Many thanks to Prof Doug Altman, Mr Geoffrey Bilder, Sir Iain Chalmers and Prof Mike Clarke for their contributions.
What is threaded publications trying to achieve?
What a reader or ‘user’ might gain from threaded publications depends on their role. As a systematic reviewer of the medical evidence, you need to easily and readily find all articles and data related to the topic you are studying. Research funders and ethics committees need to know if a new piece of research, such as a clinical trial, is warranted (indeed, all clinical trials should ideally not begin without a prior systematic review of the evidence). Healthcare practitioners and patients might want to find as much information as possible that will help them make well-informed choices about practice. And as open access publishers, we want to provide a service to authors that helps them to enhance the healthcare literature to increase the visibility, discoverabilty and pace of research.
But the aim can be captured more succinctly. In short, in healthcare, we want science to be more efficient, to improve patient care, and to produce better outcomes for people and communities.
Other considerations
Prevention of publication bias
The publication of all sound scientific results and sharing of data is needed to ensure the availability of unbiased evidence about the effects of treatments. Yet another high-profile example (statins proven ineffective for atrial fibrillation) emerged last month. Blame is sometimes laid on funders of research (with the pharmaceutical industry being a common target) but many stakeholders in the research funding, peer review and publication process – albeit, inadvertently – are contributing to this problem of emphasising the positive and hiding the negative.
BioMed Central publishes several online peer-reviewed journals that specifically aim to publish negative, inconclusive or small scale studies, which might not find their way into highly selective or space-restricted journals. Through journals such as Journal of Negative Results in Biomedicine, Trials, and BMC Research Notes researchers should be able to more easily find the available evidence on a particular treatment. Threaded publications and increased data sharing will take this to another level.
Legal and ethical requirements
Clinical trialists should register their trials in internationally recognized databases before submitting their research to their chosen journal. This is an ethical, journal editorial policy and, increasingly, legal requirement. And, for federally-funded researchers in the USA, reporting of summary trial results in clinicaltrials.gov is required. ICMJE guideline-endorsing journals are obliged to publish negative results. However, it is clear restrictive duplicate publication policies and lack of interest in negative or inconclusive results by some journals makes compliance difficult.
Defining and developing ‘the thread’
Scientific research is a circular process; we inherently build on previous knowledge to form new hypotheses, gather data and publish results to draw conclusions, which then inform subsequent research. Threaded publications are more pyramidal than circular. A threaded publication should be considered more in the context of a particular trial of a particular treatment, linked by a unique identifier – such as trial ID or ISRCTN.
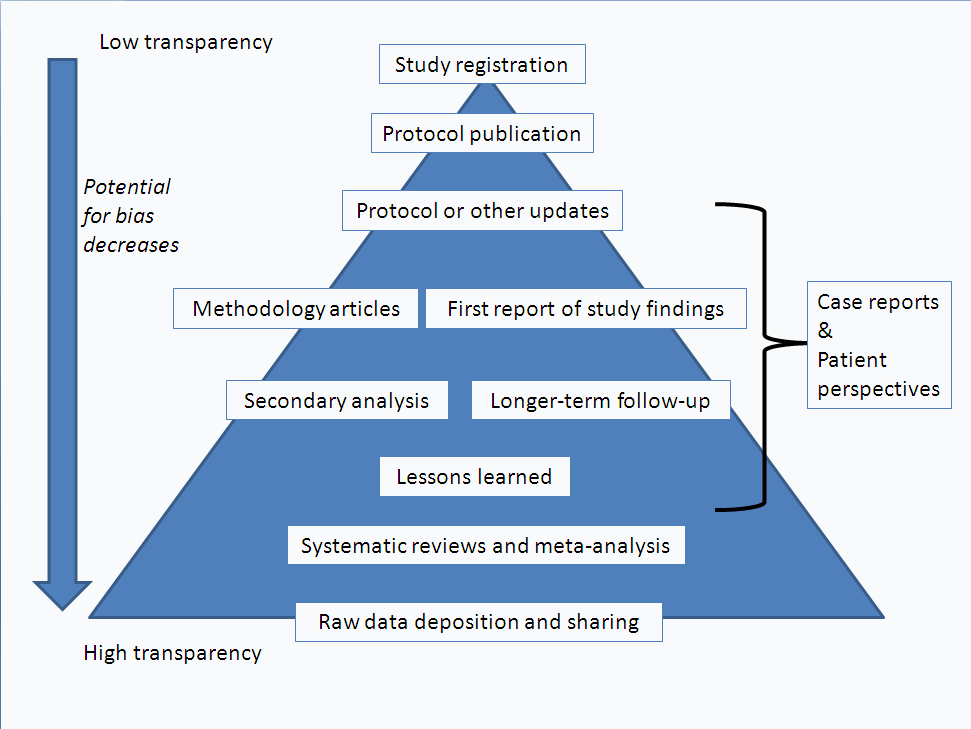
As you progress through a clinical trial, by sharing (publishing) more information you arguably increase transparency in the literature and reduce the potential for bias.
Next steps
The response to our initial threaded publications announcement has been pleasing but there is much more of the concept to develop.
While the publication of all elements of a thread within BioMed Central’s publishing services will enable maximum value to be added to a sequence of publications in the longer term, the concept of threaded publications should fundamentally span the literature, and multiple publishers. We are therefore pleased to be working with CrossRef to develop a proof of concept for threaded publications which we hope will eventually be interoperable between journals and publishers. CrossRef is the independent association founded by publishers which issues Digital Object Identifiers (DOIs) to scholarly works to ensure permanence – assigning a DOI to an article means that if information about the article changes, such as its URL on the internet, it can still be found.
The foundation of threaded publications – and science – are data. However, due to many challenges associated with openly sharing clinical research data there is a paucity of open access clinical trial data in the literature. But there are journals willing to consider publishing peer-reviewed articles about published data sets. So to further help us prove the threaded publications concept we encourage any author of a clinical trial published in a BioMed Central journal to contact us to discuss submitting their data set for consideration by Trials or BMC Research Notes as part of our proof of concept (we will waive the article processing charge if it is accepted).
Twitter hashtag: #threadedpublications
Comments