Breast cancer is the most common cancer to affect women. Each year it causes the death of over 500,000 people. However, it is not very well understood how breast cancer starts and how it grows into abnormal tissue.
There are many similarities between the ways that cells behave in normal breast development and in cancer. Ideally one would like to have suitable laboratory models of normal breast development and function, in order to explain what goes wrong during cancer formation and progression.
3D culture models
Tissue culture dishes are not ideal because the cells isolated from tissues end up being stretched out in sheets and their local environment in culture is very stiff.
The use of cultured cells is crucial for explaining how animal biology works. Unfortunately tissue culture dishes are not ideal because the cells isolated from tissues end up being stretched out in sheets and their local environment in culture is very stiff.
Many tissues are soft, and the cells are always arranged in an intricate 3-dimensional architecture. So on culture dishes, cells isolated from tissues frequently loose their normal function and quite often they die. However, soft 3D culture models change this picture dramatically.
We’ve known for a couple of decades that under these 3D conditions, cells can form tissue-like structures. Breast cells also make the specialised proteins they are supposed to make, such as milk proteins, and they last quite a long time as well.
Breast tissue architecture
Breast tissue consists of a branched network of hollow tubes lined with ‘luminal’ epithelial cells, which are surrounded by ‘myoepithelial’ basal cells. Outside of this, the ‘stromal’ cells make the surrounding structure of the tissue, which provides it with solidity. When a mother gives birth, some of the luminal cells form into spherical alveoli, which make the milk. The myoepithelial cells then squeeze milk into the ductal tubes, which transport it to the nipple for the baby.
New research has now developed novel types of 3D scaffold that allow human breast cells to form and expand into intricate tissue-like ductal structures, similar to those found in the body. Indeed, in this work, artificial gels have now been constructed that mimic the breast tissue environment.
What was carried out?
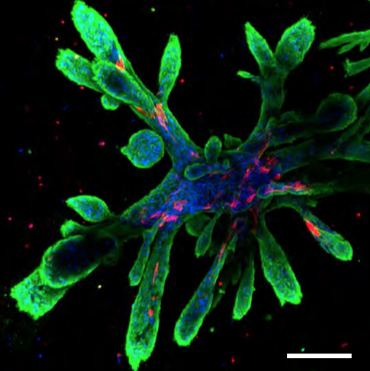
Breast cells were isolated from women undergoing reduction mammoplasty. They were then added to a mixture of extracellular proteins called laminin and fibronectin, together with hyaluronan and growth factors (insulin, epidermal growth factor, hydrocortisone).
Collagen was added to form a solid gel. The cells were then left at 37ºC to grow and to elongate into ducts for 2 weeks.
The cells organized themselves and formed branched structures similar to those in the tissue. Human breast cells have not previously been grown in this way before, and the cultures that emerged are quite striking in appearance.
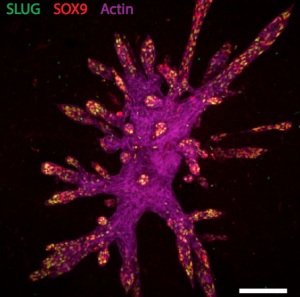
The 3D scaffolds allowed the cells to form tubular structures similar to those in the breast, with different groups of cells expressing markers of each cell type and the cells were orientated correctly. Moreover the tubes also showed hormone sensitivity. In fact they could hollow out in response to milk-stimulating hormones thereby allowing milk flow, and they could make milk fats.
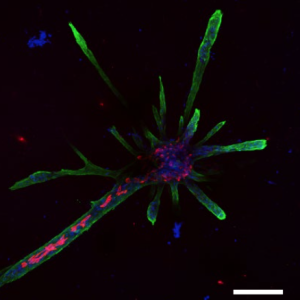
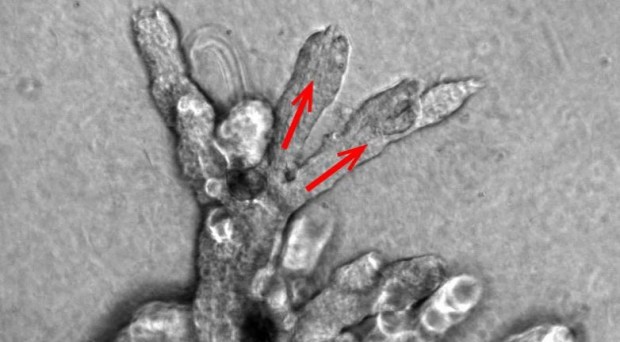
The work also explored how branches form. It was previously thought that luminal cells drive branching by exerting an expansive force on the tip of the branch. However, it now seems that one or two so-called ‘leader’ stem cells at the tip of the branch reach out to the surrounding tissue and remodel it to facilitate branch elongation.
What are the implications?
With this new culture technique, researchers will be able to use it to explore how the formation of breast tubes and the development of a lactating breast really occur.
With this new culture technique, researchers will be able to use it to explore how the formation of breast tubes and the development of a lactating breast really occur. Importantly, they will also start using it to understand much more about what goes wrong in breast cancer, and how the structure of the tissue becomes abnormal in the disease.
In our own work, we are interested in breast density (which can be seen in mammograms), the associated changes in mechanical stiffness of the tissue, and how this might lead to cancer. So the model may also help us to determine what goes wrong in breast cancer, and how increased extracellular stiffness within this tissue increases the risk of getting the disease.
Comments