These questions have occupied the thoughts of neuroscientists over the years and formed the subject of many separate investigations. Roughly a third of the fluid is located in cavities within the brain or in spaces surrounding the brain where it cushions it inside the skull so playing a vital role in protecting it against mechanical trauma. The other two-thirds of the fluid is located within the brain in the very fine spaces separating and surrounding the brain cells. By perfusing the cells it is important in assisting delivery of nutrients and in removal of waste products. It also provides the correct environment to allow the brain cells to function efficiently, independently of any fluctuations occurring in the general circulation.
It has been known for many years that between this brain fluid and blood there are barriers that limit and regulate entry and exit of many substances. These key interfaces are the choroid plexuses that protrude into the brain cavities and the blood-brain barrier provided by the lining of the blood vessels that deliver blood to all parts of the brain. However the relative roles that the choroid plexuses and blood-brain barrier play in regulating the amount and composition of the fluid within the brain is still not well defined.
A comparative account of mechanisms and roles
In our latest review we survey the literature on the subject in the hope of clarifying the extent to which each of the interfaces produces the fluid and regulates the amounts of the most prevalent components e.g. the water and salts. The diagram below indicates fluid movements into and out of the brain across these interfaces.
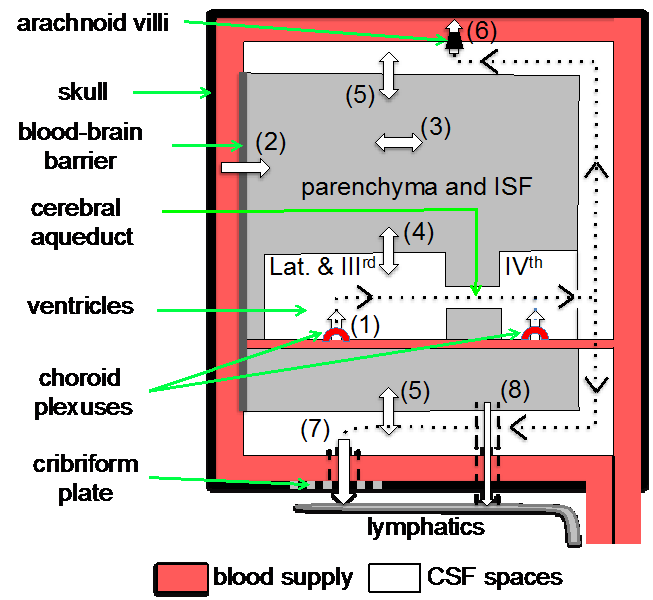
We highlight the hazards and difficulties in interpreting the results and point to misconceptions that have crept into the literature because of the difficulties of measuring overall net movement across the barriers of major components of the fluid e.g. sodium and chloride. Our review also emphasises that movements of radiotracers for sodium and chloride ions across the blood-brain barrier in each direction have been found to be so much larger than any difference that might exist between them that measurements of tracer movements cannot be used to calculate any overall net movement either net secretion into or net absorption out of the brain.
Measuring ion and fluid transport at the blood-brain barrier
Many of the early studies on sodium and chloride movements across the blood-brain barrier used specific inhibitors to try to determine the mechanisms but failed to find any measurable effect on the large unidirectional movements of these ions. These results have been said to demonstrate that there is no net movement of fluid across the blood-brain barrier. We argue that most of the tracer movements occur by a mechanism not affected by inhibitors that has large and almost equal amounts moving in and out of the brain. However there is a small portion of the tracer movements that occurs by a different mechanism that is susceptible to inhibition and that can discriminate between inward and outward movements so leading to fluid secretion into or absorption from the brain. The lining of the blood vessels that make up the blood-brain barrier possess the transporters needed for such a mechanism and these transporters are affected by the inhibitors.#
We also argue that it is the choroid plexuses that produce most of the fluid but that the blood-brain barrier is responsible for adjusting the degree of acidity (the pH) and the potassium ion concentration in the fluid both factors having a profound influence on the excitability of nerve cells in the brain and thus being crucial to normal brain functioning. In particular we discuss how the bicarbonate transporters that we have identified at the blood-brain barrier are important for adjusting the acidity in the brain. We evaluate the evidence showing how much bicarbonate is transported and how fast this occurs. The conflicting evidence is summarised in the table below.
![Summary of evidence that has been used to support or oppose rapid transport of HCO3 following changes in [HCO3 ]plasma at constant pCO2) Based on this re-examination of all the evidence we argue that bicarbonate transport is slow, comparable to that of other ions, but nevertheless it is still fast enough to be the major determinant of bicarbonate concentration in the brain.](https://blogs.biomedcentral.com/on-biology/wp-content/uploads/sites/5/2016/12/barrandhladky2.png)
At the end of our review we provide a summary table that neatly sets out and compares the features of the two barriers and in the concluding section we discuss important areas of uncertainty and provide pointers to future investigation. These include: direct measurements of net fluxes across the blood-brain barrier; recordings of intracellular potentials during transport across the blood-brain barrier and the choroid plexuses under conditions close to those obtaining in vivo; localization of transporters at the blood-brain barrier to the luminal or abluminal membranes in particular the distribution of Na+-pump activity and whether this can be altered in response to different challenges.
Steve Hladky and Margery Barrand
Margery Barrand is a retired Senior Lecturer in Pharmacology at Cambridge University who brings a more cell-biological approach to bear on the interpretation of results and has extensive research experience of the transport processes that are located at the blood-brain barrier, especially the multi-drug transporters (sometimes called foreign molecule or xenobiotic transporters) that provide protection to the brain.
Comments