
Today is World NTD Day. Committed Bugbitten followers will know that our blog posts are normally published on Fridays, however this week we decided to align ourselves with this global campaign to highlight Neglected Tropical Diseases (NTDs), the majority of which are caused by parasitic infections. Research and technological development are cornerstones to the global effort to eliminate NTDs.
Infectious Diseases thriving on inequities
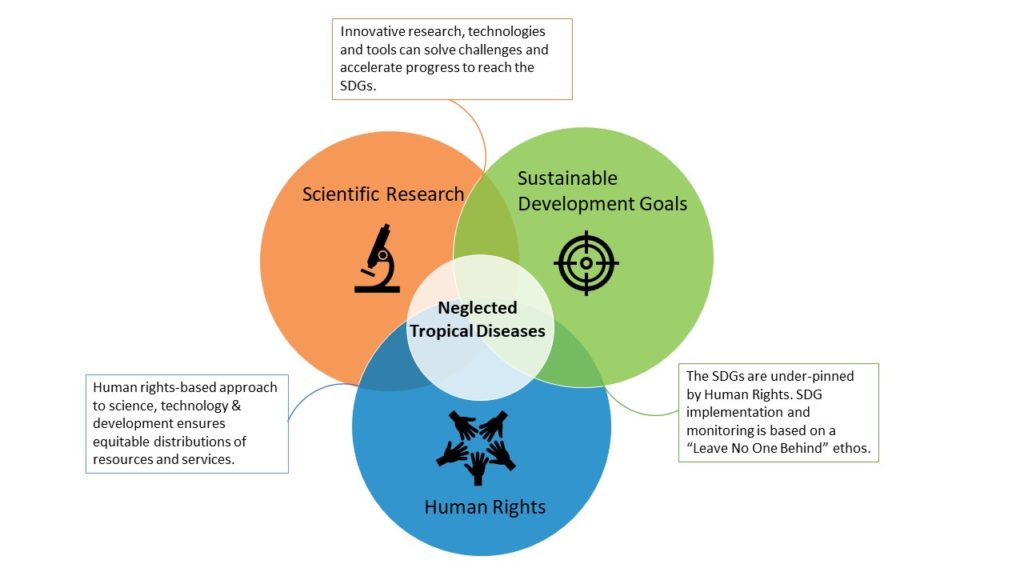
NTDs sit in the middle of a Venn diagram, where scientific research, sustainable development and human rights meet. Predominantly affecting people living in deprived, underserved and disenfranchised communities, NTDs succeed through our failure to correct inequities. NTDs both thrive in areas of poverty and strengthen the causal roots of poverty, by stunting child development, robbing productivity, reducing quality of life and causing irreversible damage and disability.
It is estimated that 1.5 billion people are affected by NTDs and 1 in 5 people are at risk of an NTD worldwide. The fight to stop these diseases has led to a global commitment (called the London Declaration 2012) by governments, multi-lateral organizations, philanthropic foundations, industry and research groups to work together to alleviate suffering and improve health and wellbeing, as highlighted by the World NTD Day motto, Beat NTDs: For Good. For All.
There have been huge successes since the London Declaration in 2012. To highlight a few: 31 countries have eliminated at least one NTD to date, over 1 billion people have been treated for at least 1 NTD, the number of people requiring treatment is consistently falling and silos are breaking down as cross-sector interventions, particularly between NTDs, WASH (Water, Sanitation and Hygiene) and Education are being implemented.
But challenges remain and it is to the scientific community that we turn to for solutions to many of these pressing public health challenges. Here are a few examples focused on recent schistosomiasis studies.
Diagnosis for all settings
The gold standard diagnostic test for schistosomiasis uses egg-based microscopy techniques as indicators of infection and as a proxy to morbidity (the higher the infection intensity, the higher the morbidity). Ongoing interventions are very effective at reducing infection intensities and thus egg-output. This means the diagnostic tests we rely on are not sensitive enough in low endemic areas, limiting their effectiveness in monitoring schistosomiasis disease elimination programmes, and rendering them unusable for transmission interruption and surveillance.
In clinical settings, complex morbidities due to schistosomiasis such as female genital schistosomiasis, male genital schistosomiasis and hepatosplenomegaly are often egg-negative and subtle morbidities are present when egg-production is not apparent.
Diagnostic tests that are sensitive, rapid, non-invasive, and that can be used at point-of-need and in resource-poor settings, are urgently needed. A recent article by Rostron et al 2019 evaluated the use of recombinase polymerase amplification (RPA), a rapid, portable, point-of-need molecular based technique (see this post) for the detection of S. haematobium that shows promising results. Research and development into diagnostics for NTDs is of vital importance.
Access to preventative treatment
Schistosomiasis control and elimination programmes rely on school-based, mass administration of preventative chemotherapy (regular treatment of infections to prevent the development of disease). There are challenges on equitable treatment access and whether as a public health programme treating entire communities or only school children is beneficial and cost-effective (Secor et al 2019 and Toor et al 2019). A different challenge is that praziqauntel, the only drug available against all schistosome species, is only licensed for over 5 year olds, the Pediatric Praziquantel Consortium is working on a new formulation for infants and toddlers.
Evaluating schistosomiasis programmes
A recent research publication by Deol et al 2020 used multi-country and multi-year epidemiological data from 10 schistosomiasis control programmes to evaluate World Health Organization (WHO) programmatic guidelines and assess if and how quickly countries reached the WHO set targets of ‘Morbidity control’ and ‘Elimination as a Public Health Problem’. Deol et al 2020 data analysis showed that, with the exception of S. haematobium in Niger, all country programmes reached the ‘morbidity control’ target after at least two rounds of treatments, much faster than the WHO timeline of 5-10 years.
The second target ‘elimination as a public health problem’, was reached by 5 countries for S. mansoni, but none of the countries reached it for S. haematobium. For S. mansoni countries with low endemicity levels reached the ‘elimination as a public health problem’ target sooner than the WHO guidelines propose.
These findings can be used when developing updated treatment guidelines and protocols for monitoring and evaluation, as well as programme progression planning.
Future proofing achievements, sustaining schistosomiasis control and elimination
A challenge of these preventative chemotherapy (PC) intervention programmes is that PC does not prevent reinfection and gains made by these interventions are jeopordised by the risk of infection prevalence persistence and resurgence. A recent study by Walker et al 2019 explores how predictors of persistent infections are linked to environmental and behavioural factors. There are research projects underway to see how integration of public health interventions programmes such as Water Sanitation and Hygiene (WASH), risk reduction through Behaviour Change interventions combined with treatment interventions, may achieve sustainable elimination of schistosomiasis and lead to interruption of transmission, for example the Geshiyaro project (covered in a previous blog post) and the WISER project.
Aquatic snails are the intermediate hosts of schistosomes, and they act as little amplifiers of schistosomiasis transmission in the water. Controlling the snail intermediate host could be highly beneficial to sustaining schistosomiasis control and elimination achievements.
A recent review explores how the use of gene drives, a genetic engineering technology that spreads particular traits throughout populations, could be used to reduce transmission in selected sites, either by spreading genes that reduce snail host populations or by spreading genes that decrease snail susceptibility to schistosome infections. Investigating which traits and hence genes could be used for snails to be resistant to schistosome infections will aid in developing such technologies (e.g. de Melo et al 2020 and Castillo et al 2020).
Our over-reliance on one drug for the treatment of schistosomiasis is a significant risk. New alternatives or combinations with praziquantel as described in this article by Sebastian Perez et al 2019 are crucial to mitigate against any drug-resistance developing to our only effective treatment against all species of schistosomes.
From informing public health decisions, improving operational and programmatic strategies to developing new technologies, the global NTD community relies on scientific research to help accelerate NTD control and elimination. Investment in research and development is crucial to maintain and improve on the huge gains made.

Comments