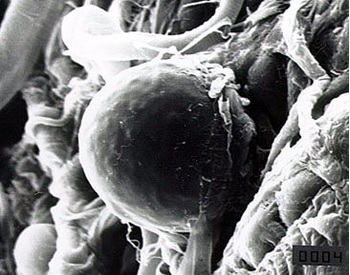
Malaria is a parasitic disease caused by Plasmodium parasites that are transmitted to their mammalian hosts by the bites of infected female Anopheles mosquitoes. Strategies aimed at controlling malaria include efforts towards blocking this crucial step of the parasite’s life cycle.
This can be achieved by measures targeting the mosquito through the use of insecticides, or by strategies aimed at preventing the parasite from developing inside its mosquito vector, a multi-step process known as sporogony. But what if both could be done at the same time?
Ivermectin is a member of the avermectin family of compounds, a class of macrocyclic lactones with insecticidal and antiparasitic properties. Ivermectin is the best-studied member of this family, and has been considered one of the most successful discoveries in the fight against infections caused by roundworm parasites.
The importance of this drug is well illustrated by the award of the 2015 Nobel Prize in Physiology or Medicine to Satoshi Ōmura and William C. Campbell “for their discoveries concerning a novel therapy against infections caused by roundworm parasites”.
Mass drug administration (MDA) of ivermectin in Africa and Latin America led to a reduction of onchocerciasis, as well as of lymphatic filariasis and scabies, which are also endemic in India and Southeast Asia. The impact of ivermectin on insect vectors, in particular its activity against Anopheles mosquitoes, further accounts for its potential as an integrated tool for malaria control. However, although ivermectin’s impact on Plasmodium transmission is well documented, whether this is solely due to its insecticidal effect or, additionally, results from its ability to inhibit the parasite’s development inside the mosquito remained unresolved.
Finding an answer to this question remained, until recently, a significant challenge, due to the inherent difficulties in extricating the effect of a given compound on Plasmodium sporogony from that it exerts on its mosquito vector.
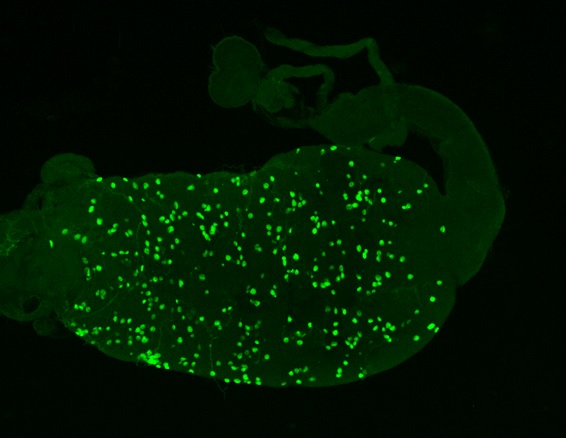
A major step towards solving this problem was made by our recent disclosure of a bioluminescence-based method to monitor Plasmodium sporogony in vitro. Briefly, this novel methodology employs a transgenic Plasmodium parasite that expresses the luciferase gene, in combination with parasite culture conditions that enable the development of its mosquito stages in the absence of… mosquitoes. In this way, we are able to assess the impact of a given drug on Plasmodium sporogony, independently of that compound’s effect on the mosquitoes.
Thus, we set out to employ this new experimental approach to answer the fundamental question of whether the inhibition of Plasmodium sporogony by ivermectin contributes to its ability to block parasite transmission. To this end, our luciferase-expressing parasite was cultured under the conditions described in the 2017 paper and incubated with ivermectin and other members of the avermectin family at various stages of Plasmodium sporogony. Our results show ivermectin, as well as other avermectins exert a strong inhibitory effect on the generation and development of Plasmodium oocysts (as shown in the picture), a key step of the parasite’s sporogonic development process.
Our results support the notion that, besides their mosquitocidal effect, avermectins may also directly target the parasite’s sporogonic stages, which likely contributes to transmission blocking activity.
Our data lend further support to the use of avermectins for MDA as a tool for malaria control in endemic regions, and suggest that the inclusion of members of this family of avermectin compounds besides ivermectin in these interventions should be considered.

Comments